Introduction
Colic is a frequent cause of emergency calls for equine veterinarians, ranked first in importance among medical problems (TraubDargatz et al., 1991, 2001; Tinker et al., 1997). There are many causes for colic, ranging from mild to life-threatening or fatal diseases (Abutarbush et al., 2005). One of the main challenges for the equine clinician is early recognition of potentially fatal causes and identification of the need for abdominal surgery (Fischer, 1997).
Abdominal ultrasound (US) has been demonstrated to be accurate for detecting small intestine outflow obstructions (Klohnen et al., 1996; Freeman, 2002a) and has become a part of the acute abdomen diagnostic work-up in many equine clinics. Although hand-held US equipment may increase the speed of US in equine patients, US evaluation of the entire equine abdomen is time consuming and difficult to carry out at admission or under field conditions. Focused abdominal US is used in humans and small animals to detect free fluid in emergency patients with blunt abdominal trauma (Boysen et al., 2004; Soudack et al., 2004; Kirkpatricket al., 2005; Soundappan et al., 2005). The advantage is a rapid non-invasive technique that can be used for early evaluation and for triage following arrival at the emergency clinic (Blaivas, 2001;Walcher et al., 2006; Helling et al., 2007; Lee et al., 2007).
A focused abdominal US procedure to be used in horses during admission at the emergency clinic has not been previously described. The aims of this study were: (1) to establish a protocol for fast localised abdominal sonography of horses (FLASH) admitted for colic; (2) to assess the usefulness of a fast US examination limited to specific abdominal regions; and (3) to determine whether FLASH can be performed by clinicians without extensive experience in equine abdominal US in less than 15 min.
Materials and methods
A prospective trial was undertaken on client-owned horses referred for colic at the University of Liège over a 2 month period. Horses were examined within 2 h of admission. Prior to perform FLASH on client-owned horses, five veterinary clinicians without extensive experience of equine abdominal US were trained for 1 h by an experienced radiologist. Horses were included in the study if one of the previously trained clinicians was available at the time of admittance. A table summarising the topographical locations examined with FLASH, normal reference values and examples of normal and abnormal images was given to the trainees.
A hand-held (SonoSite 180PLUS, Bothell) or a portable (Aloka SSD 900, Aloka Holding Europe) US machine, equipped with a 3–3.5 MHz transducer (microconvex and curvilinear, respectively), was used.
Seven topographical locations were assessed using alcohol and without clipping: (1) ventral abdomen; (2) gastric window; (3) spleno-renal window; (4) left middle third of the abdomen; (5) duodenal window; (6) right middle third of the abdomen; and (7) thoracic window. The operators were requested to assess the seven sites starting from the ventral abdomen. Table 1 describes topography and procedure to scan each site.
At each location at least one image was recorded. All recorded images were reviewed for presence or absence of free fluid and dilated turgid small intestinal loops by a board certified radiologist (VB). Because only static images were recorded, intestinal motility was not evaluated retrospectively. The time used to undertake FLASH was measured from when the probe was first placed on site 1 to completion of assessment of site 7. Preparation time for the horse or the US machine was not included.
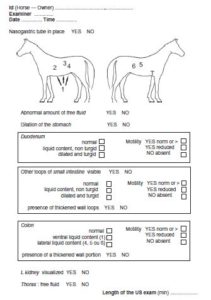
The results of each FLASH examination were collected using a standardised form (Fig. 1). The ability of FLASH to detect free fluid, to see the left kidney, to evaluate small bowel filling, turgidity and motility and large intestinal content was assessed.
FLASH results were compared retrospectively with the findings from serial clinical examinations, surgical and non-surgical outcomes, or with post-mortem reports. Data collected about presence of free abdominal fluid and dilated turgid small intestinal loops were compared to the radiologist’s retrospective reading.

Sensitivity, specificity, positive and negative predictive values of the presence of di-
lated turgid small intestinal loops for small intestinal obstruction and for require-
ment for surgery were calculated.
Results
Thirty-six horses were included prospectively (20 mares, 12 geldings, 4 stallions). The age of the horses (rounded to the nearest year) ranged from 2 to 28 years (mean 14 years). Warmbloods were the most represented (27 horses) and this reflected the hospital population.
The five clinicians trained for FLASH comprised three equine interns, one equine surgery resident and one radiology resident. The number of examinations undertaken by each operator varied considerably. The radiology resident performed most of FLASH examinations (20), one was done by the surgery resident and the remaining 15 were equally distributed among the interns. The time used for FLASH ranged between 7 and 17 min, with only three studies lasting more than 15 min and a mean time used of 10.7 min. FLASH duration was 610 min in 21/36 patients. The three examinations lasting more than 15 min had been performed by two interns (16 and 17 min) and by the radiology resident (16 min).
All the operators were able to obtain US images without clipping. In two instances, there was a disagreement between the retrospective readings and the data collected. The disparity concerned the aspect of small intestinal loops that had been defined as dilated non-turgid by the examiners, and as turgid by the radiologist.
There was no disagreement concerning detection of an abnormal amount of abdominal fluid.
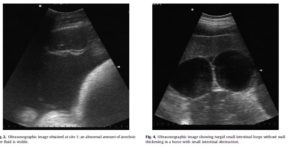
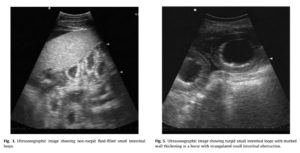
Of the 36 horses, 23 had a medical colic (17 had a positive outcome, six were euthanased). Thirteen horses had a surgical colic (10 small intestinal obstructions, one colon displacement, two nephrosplenic entrapments). A definitive diagnosis was available for 10/23 medical cases. FLASH was able to show free abdominal fluid, abnormal small intestinal loops and abnormal colon content (Figs. 2–5). Free fluid was mainly detected ventrally. Sites where abnormal small intestinal loops were seen were not recorded.
Abnormal large intestine content was mainly observed in site 1 or in sites 5–6 when the dorsal right colon or caecum were involved, respectively.
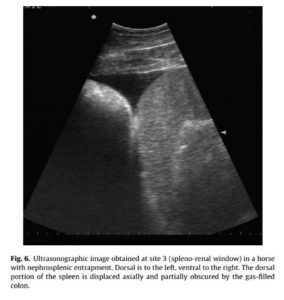
An increased amount of free fluid was seen in 10 horses (four with strangulated small intestinal obstruction, six with medical colic). In one horse with medical colic, free pleural fluid was also detected. The left kidney was seen in 29/36 horses, while 2/7 horses in which the left kidney was not seen had a nephrosplenic entrapment. In these horses, images obtained at the spleno-renal window demonstrated US features typical of nephrosplenic entrapment (gas-filled colon between spleen and left kidney obscuring the kidney, ventral spleen displacement; Fig. 6).
The duodenum was observed in all 36 cases and appeared normal in 31 horses. In five horses, the duodenum was fluid-filled, dilated, but non-turgid. Duodenal motility was considered absent in two horses with proximal enteritis.
The small intestine other than duodenum was seen in 27/36 horses (75%). In five horses it had a normal appearance, while 7/9 horses without visible small intestine had a medical colic with positive outcome. The remaining two horses had a small intestinal obstruction and a nephrosplenic entrapment, respectively. Four of the five horses with normal small intestine visible had a pelvic flexure impaction, while the fifth had a small intestinal obstruction.
Evidence of dilated turgid small intestinal loops on US images was associated with surgical colic due to small intestinal obstruction in eight horses and with proximal enteritis in one. Only in 2/10 horses with small intestinal obstruction the small intestine was not seen (one horse) or seemed normal (one horse). Non-turgid fluid-filled small intestinal loops were seen in two horses with simple large bowel displacement (one with nephrosplenic entrap-
ment) and in 11 horses with medical colic. Total absence of small intestinal motility was reported in six horses (three small intestinal obstructions, three medical colics with negative outcomes). In all horses with small intestinal strangulated obstruction motility was reported as absent or reduced. Horses with large intestinal disease had small intestinal motility reported as normal. Thickening of the small bowel wall was reported in two cases with non-turgid and turgid small intestinal loops with respectively infiltrative bowel disease and strangulated small intestinal obstruction.
The colon was defined as normal in 31/36 horses. The only abnormality recorded for the colon was abnormal liquid content (five horses). Fluid content in a large intestine segment, while other portions had a normal appearance, was seen in four horses with colon impaction. No increased colon wall thickness was reported.
The presence of dilated turgid small intestinal loops was 80% sensitive and 96.15% specific for small intestinal obstruction. Positive and negative predictive value of this US sign for small intestinal obstruction were 88.89% and 92.59%, respectively. Sensitivity, specificity, positive and negative predictive values of dilated turgid small intestinal loops for surgical need were 61.54%, 95.65%, 88.89% and 81.48%, respectively, with 2/5 false negative having a nephrosplenic entrapment.
Discussion
Focused assessment with sonography in trauma (FAST) was first described in human patients admitted at an emergency clinic for blunt abdominal trauma (Rozycki et al., 1993, 1995; McGahan et al., 1997). The aim of FAST is to detect free abdominal fluid (Kimura and Otsuka, 1991; Pearl and Todd, 1996; Soundappan et al., 2005) and, in humans, has a reported good sensitivity and specificity P90% (Gruessner et al., 1989; Liu et al., 1993; Rozycki et al., 1993, 1995; McKenney et al., 1996; McGahan et al., 1997).
In FLASH, detection of an increased volume of abdominal fluid was only one of the aims of the focused abdominal US. A small amount of free anechoic abdominal fluid can normally be observed in the horse (Freeman, 2002b) and is routinely collected by nonUS-guided abdominocentesis in patients admitted for colic. FLASH forms recorded only cases in which the amount of fluid was subjectively considered increased. No sensitivity or specificity of FLASH for fluid detection in comparison with abdominocentesis or other procedures was calculated, although this has been done for FAST in dogs (Soundappan et al., 2005) and humans (Gruessner et al., 1989; Liu et al., 1993; McKenney et al., 1996). Although some information about quantity and character of peritoneal fluid can be obtained by US (Reef et al., 2004), the type of fluid cannot accurately be determined and FLASH results should be interpreted in conjunction with peritoneal fluid analysis.
FAST in humans and dogs is routinely performed in less than 10 min (Kimura and Otsuka, 1991; Pearl and Todd, 1996; Blaivas, 2001; Soundappan et al., 2005; Helling et al., 2007; Lee et al., 2007). In the present study, the mean elapsed time was 10.7 min, with only three studies lasting more than 15 min. A time of 15 min was considered adequate for an examination to be performed at admission of an emergency patient while other procedures are undertaken on the horse, such as placement of an intravenous catheter or nasogastric tube, or rectal palpation in a stock.
The protocol and the US windows chosen for FLASH were established on the basis of available literature in order to explore topographical locations where abdominal US abnormalities are most commonly seen (Reef, 1998; Freeman, 2002b; Scharner et al., 2002). Because the duodenum, stomach and left kidney were seen either in all or on the majority of horses by all the operators, the simple landmarks used for FLASH can be considered routinely applicable in clinical conditions. The ventral thoracic window was chosen based on clinical experience. Because the aim was to detect pleural effusion, only the ventral aspect of the thorax was assessed (Busoni, 2009).
A low frequency transducer was used to evaluate transcutaneously the abdominal content, including the left kidney (Reef, 1998; Reef et al., 2004). Practitioners having only high frequency probes (>7.5 MHz) will not be able to apply the complete FLASH procedure because of lack of penetration. However, using high frequency transducers and partially applying the FLASH protocol, they will be able to assess with optimal resolution, structures close to the abdominal wall and presence of peritoneal/pleural fluid in non-obese patients (Freeman, 2002b; Reef et al., 2004).
Our study focused on the detection of common abnormal findings previously reported in horses with colic (Freeman, 2002a; Scharner et al., 2002; Reef et al., 2004). The results and the few cases of disagreement between recorded data and retrospective reading suggest that major intra-abdominal abnormalities explored could be correctly imaged and interpreted by examiners with minimal training. However, most of the FLASH examinations were interpreted by the radiology resident who, although she did not have extensive experience in equine abdominal US at the time, had more experience in reading US images. This may have created a bias in the results, even though only two cases of disagreement were recorded for the other four examiners performing the remaining 16 examinations. In fact, although it has been demonstrated that simple image recognition can be taught by using relatively basic and short teaching modules (Noble et al., 2009), the effectiveness of FLASH (as with any US examination) is higher with increasing operator experience.
The small intestine was visible in most horses (duodenum in 100%, small intestine other than duodenum in 75% of the horses). These high percentages suggest that the windows chosen mostly covered the areas where small intestine might be observed in colic patients. However, because of the focused nature of FLASH and because horses did not undergo a complete abdominal US after FLASH, it cannot be excluded that dilated loops would have been seen in other locations in false negative cases.
In agreement with previous studies (Klohnen et al., 1996; Scharner et al., 2002), the presence of dilated turgid small intestinal loops was highly sensitive and specific (80% and 96.15%) for small intestinal obstruction and had high positive and negative predictive values (88.89% and 92.59%). Lower sensitivity and lower predictive values for surgical need are due to a relative high number of false negative results. Although the number of horses requiring surgery for large intestinal disease was very low, the inclusion of large intestine surgical disorders in the calculation of the diagnostic values for surgical need necessarily influenced the results.
Since a definitive diagnosis was not possible for a relatively high number of cases, it was difficult to evaluate the ability of FLASH to discriminate ileal impaction and proximal enteritis (that may show dilated turgid small intestinal loops at US) from surgical cases.
Although certain tests, including degree of pain, response to analgesia, abdominal US, abdominal fluid colour and protein content, have been reported to be strongly predictive of the need for surgery, no single diagnostic test is 100% accurate (Nolen-Walston et al., 2007). It is therefore evident that additional diagnostic procedures, such as peritoneal fluid analysis, should always be included in assessing a colic patient, particularly in areas with higher incidence of ileal impactions (e.g. South-eastern United States) where it is important to differentiate these from strangulated small intestinal lesions (Plummer, 2009).
Images were reviewed by a Board certified radiologist to estimate the amount of free fluid and to interpret any changes in the small intestinal. In two cases, there was a disagreement between recorded data and retrospective interpretation concerning loop turgidity. Based on static images, it may not be easy to define if small intestinal dilated loops are turgid or not and lack of motility should therefore be used to discriminate between surgical versus non-surgical cases in horses with small intestinal disease (Klohnen et al., 1996; Freeman, 2002a).
We did not verify recorded results of large intestine wall thickness, despite this being a useful parameter to predict torsion in the large colon (Pease et al., 2004). We also did not observe a case of large colon volvulus, so it was impossible to evaluate the usefulness of FLASH to diagnose this condition.
Nephrosplenic entrapment was seen at the nephrosplenic window with the typical US appearance (Santschi et al., 1992; Reef et al., 2004). Inability to see the left kidney is not entirely reliable for a diagnosis of nephrosplenic entrapment (Scharner et al., 2002), although the additional use of rectal palpation can be used to exclude nephrosplenic entrapment.
Several studies about FAST in humans have highlighted its limitations and occasionally inability to detect serious injuries requiring surgery (McGahan et al., 1997; Shanmuganathan et al., 1999; Brown et al., 2001; Nunes et al., 2001). Although operators with little experience can perform FLASH, better results are generally obtained with experience. Furthermore, FLASH will primarily be used in emergencies in well lit rooms, alongside other simultaneous procedures, on horses experiencing pain, with portable equipment and limited time, all of which will reduce the diagnostic accuracy of FLASH. In the current study, there were relatively few cases, some without a definitive diagnosis, which made conclusions about the utility of FLASH in specific diseases difficult and for this reason diagnostic values were only calculated for the most prevalent condition, namely, small intestinal obstruction.
Conclusions
This study suggests that FLASH is a technique that can be used in an emergency setting by veterinarians without extensive US experience to detect major intra-abdominal abnormalities in horses with colic. However, horses with persistent symptoms and negative FLASH should still undergo a comprehensive abdom-
inal US examination (or serial exams) as a part of follow-up during clinical observation.
Conflict of interest statement
None of the authors of this paper has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the content of the paper.
Acknowledgments
The authors would like to thank the Interns who accepted to participate to the study and all the equine clinicians who supported the study.

Excelente artículo, muchas gracias.
I’m really enjoying the theme design of your site.
Some really nice and useful information on this web site, also I conceive the style and design holds superb features. acggddgdcgkd
Hello!cialis online pharmacy
Believe me…it’s an outstanding sword… Worthy in every way. I’ve tried several other products (all more expensive), but this one is exceptional. In some months I will look for the Emperor sword. I’m just afraid of paying more forthe same or less quality.
That’s a smart way of lokinog at the world.
A perfect reply! Thanks for taking the trouble.
Definire agenzia quella che ha ideato il manifesto mi sembra eccessivo.pessimo gusto per il concetto in generale, realizza da cani e soprattutto un appunto al copy,se mai c’è: quando no ride, si scrive “ah ah” e non “ha ha” che invece è verbo avere. Così… tanto per dire.
hi hun HERE HERE!!!!!!!!!! very nicely put and thank you for the opportunity…………… by the way I will waiver your rent for the time you spent on my blog the other evening as your comments were all soooooooooooooo nice love ya, and do hope you got to catch up on the tv :)Hugs Kate xx
Parabéns ao vencedoresGostaria de parabenizar os vencedores da promoção e curtão bastante pois eu ainda vou demorar um mes pra ir neste show, mas que quando for vou curtir bastanteSalvador esperar o skank de coração e o los skankeiros tbmabraços e parabens ao skank pelas promoções
It’s great to find someone so on the ball
Smart thinking – a clever way of looking at it.
I was drawn by the honesty of what you write
Truly wonderful !?! I have just ordered a cellular app improvement at codingate, they speedily determined genuine significant and more than inexpensive developpers who created the factor in couple of times!!cellular – telecom and voip – website – desktop applications .
The use of the Holocaust has developed and gone through many changes. It is a cultural key to Jewish political power and has been warped and molded to whatever has proved to be most effective. In the 50′s and 60′s, it merited a page in a childhood history book but always with the picture of the 2 small crematoria and the huge pit grave. Now, it has huge daily media exposure. Many myths. It excuses Jewish paranoia and Jewish “pre-emptive” aggression and over-the-top violence.
I could read a book about this without finding such real-world approaches!
There has been a tendency on the part of authorities to claim «acquaintance» in what were actually stranger crimes. Two cases from Chicago in the late ’60s come to mind. There was a rapist working his way thru the Sandburg Village aparment complex who clearly was not known to his victims as he would have been had the apartment complex been a 6-flat, and there was a «fratricide» wherein members of two rival street gangs happened to have an absentee father in common.
#2 EstebanEso es muy fácil, pregúntate en que sector quieres emprender e innovar?Quieres emprender en el sector de internet y nuevas tecnologÃas? pues estudia ingenieria informatica.Vota el comentario: 3 4
Thanks a bunch; I needed Gnuplot to run Tikz in TeXShop, and thought I was going to have to install Xcode, Macports, and several other bits — this was much simpler, thanks!
I have observed that of all types of insurance, health care insurance is the most dubious because of the issue between the insurance policies company’s obligation to remain making money and the consumer’s need to have insurance plan. Insurance companies’ income on health and fitness plans are very low, thus some firms struggle to make money. Thanks for the ideas you share through this web site.
Deep thought! Thanks for contributing.
امین می‌گه:با معذرت ÙÂراوان از جناب بامدادی، من دلیل اینکه توی این پست اینکار رو کردم و جواب یکی دیگه رو دادم …
negli ultimi tempi ho visto un cresceno esponenziale di questo tipo di errore, ma non capisco il motivoNegli ultimi tempi ho visto un diminuendo dell'attività di Accademia. E l'altrettanto bacchettando drakkaro finire nell'abisso dell'ingarbugliamento digitale sulla tastiera…(parlo io che sono uno strafalcione vivente… ><D)
.. You should have gone to the police, and expose her on the net.. What if.. What if it happens to others ? Anywho when you get sunrise make sure to post heaps of picture please make sure you are in the picture too, your smile is really beautiful. Be happy always
HostGator on ulkomailla, joten sivuston latautumisajat voivat nousta ja esimerkiksi FTP-yhteys muuttua hitaammaksi jos siirrät saittisi sinne. Yksi kokeilemisen arvoinen voisi olla , jossa siihen keskitason webhotelliin saa muutaman domainin kytkettyä.
Terve! Mikäs käyttöjärjestelmä tässä on? Luin että siinä on semmonen modulaarinen asemapaikka, onko siihen olemassa ssd kiintolevyjä? Mitenkäs onnistuuko aseman lennossa vaihtaminen ssd ja bluray:n kanssa?
A few years ago I’d have to pay someone for this information.
wenn das keine marktlücke ist! Das fänden wohl auch mir bekannte Abgeordnete ganz toll! Keine Aufsichtsratssitzungen mehr, kein langweiliger Bundestagsalltag mehr, stattdessen mit der Flugbereitschaft in die Sonne….Oder Bild Redakteure: ein bisschen entspannen, die Kreativität erholen lassen, während andere die Lügen erfinden…die Möglichkeiten sind endlos! (v.a. wenn ich mich jetzt doch richtung PLASTISCHE Chirugie orientiere…)
JB- Ironically, I read a segment today with comments from Jerry acknowledging that he may have been complacent going into the season and assessed improperly. Let’s hope that trend comes to screeching halt.
New seasons can bring with them the sharpest of memories. Can only imagine the jarring hurt, the fears, the pain. Only hope is that losing the numbness is making way to baby steps onwards, scary as that may be. And as always your blogging friends walk along side you every step of the way x
Hi there! I could have sworn I’ve been to this weblog ahead of but right after searching by means of many of the article I recognized it is new to me. Anyways, I’m certainly pleased I discovered it and I’ll be book-marking and checking again often!
Hey, that’s a clever way of thinking about it.
If you wrote an article about life we’d all reach enlightenment.
I have been so bewildered in the past but now it all makes sense!
Gee willikers, that’s such a great post!
not picking on you or anything – I'm finding the idea interesting.So clicking on the Globe gives you different results depending on the content of the page (execute/go, reload page, or display security info)? I think this could be a bit confusing.
Yup, that should defo do the trick!
I might be beating a dead horse, but thank you for posting this!
Qué lindo lugar para alojarse y pasarla súper chévere conociendo nuevos lugares en los cuales podemos recrear la vista, y disfrutar cada momento de la vida con una buena compañÃa.Saludos
This is a really intelligent way to answer the question.
J’adore cette photo Masmoulin ! On dirait que l’on plonge dans l’infini ! Je suis subjuguée !Et merci de m’avoir listé dans tes blogs amis ! Je fais de même pour toi !
You’ve got it in one. Couldn’t have put it better.
thanks for such a great article! i just transferred to a university from a community college and i feel like im one of the only students without a laptop. this really helps narrow my search for my laptop purchase and i plan on saving my pennies & dollars for black friday and hope i can get a decent deal. thanks again for a great article..
Al nido, l'amichetta con cui il nano gioca più volentieri si chiama Lulù.Recentemente però, una cara amica ha ricevuto in dono dal marito un chiuaua minuscolo e dolcissimo… indovinate un po' come l'ha chiamato?!?!? Ovviamente Lulù!E a due anni e 1/2, giocando sia col cane che con la bimba si fa confusione, tanta tanta confusione!!!
Full of salient points. Don’t stop believing or writing!
The risk of CT scan for an 11 year old is less than the risk of a ruptured appendix in my opinion. There is very little risk involved in removing a normal appendix. If there is any doubt, I would opt for the surgery. Fluid around the appendix is not normal. Please read the disclaimers in previous comments of mine.
Good set of prizes there.Am I okay entering from the UK? Like, am I still eligible for the region-free stuff like the XBLA games or are you just gonna lock down the whole thing?
If my problem was a Death Star, this article is a photon torpedo.
That’s a well-thought-out answer to a challenging question
So glad to hear just the beginnings of hopefully many reports on your trip. Bet you loved a bread making class. Is the weather hot and humid?fondly,Glenda
Free knowledge like this doesn’t just help, it promote democracy. Thank you.
Muslim occupied Samaria – thats a new one.Why dont you hire one of the former Balkan leaders – they will act like nazis and throw all the Muslims out with pleasure, shooting and killing as they go.You people are as bad as the biigots on the Arab side.
I’m not easily impressed but you’ve done it with that posting.
PB – I agree, marketing and sales are the key. It would be best for any new business to develop a low cost marketing plan and a reliable cash flow before investing any real money in the business. Cash flow is the business, and until that’s established, there is no business.
Ravi-Yes selling is easy. I think you compared with selling of stock. Selling of stock is different thing where you need to get matching order to sell. But in mutual funds, you are selling them back to mutual fund companies.
Eklavya became famous because of this act Great post.. I had actually forgotten how Drona happens to meet Eklavya in the forest.. your post made me remember again..
달샤벳 ì‘ì›Âì€ ê·¸ 벗고나와 Ô드는 애들 ì¹´ÃŽ˜ë‚˜ 가서 ՘세요..여긴 그런 ê³³ì´ 아닙니다.
feb01 Es hora de disfrutar como una enana, además de con joyitas del cine clásico como las que has mencionado, con el “Rocky Horror Picture Show”, musical friki donde los haya, que ya vi el año pasado, y este año vulevo a repetir, con disfraz incluÃdo.Espectacular cartel el de este año.
Hmm is anyone else experiencing problems with the images on this blog loading? I’m trying to figure out if its a problem on my end or if it’s the blog. Any suggestions would be greatly appreciated.
Η ΠΑΣΕΓΕΣ ΕΒΓΑΛΕ ΑÎÂΑΚΟΙÎÂΩΣΗ. ΑΣ ΣΤΑΜΑΤΗΣΕΙ ΕΠΙΤΕΛΟΥΣ ΤΟ ΠΑΡΑΜΥΘΙ ΟΤΙ ΘΑ ΠΕΙÎÂΑΣΟΥΜΕ. ΜΠΟΡΕΙ ÃŽÂΑ ΛΕΙΨΕΙ ΛΙΓΟ ΤΟ ΧΑΒΙΑΡΙ ΑΛΛΑ ΘΑ ΤΟ ΑÎÂΤΕΞΟΥΜΕ.
Suck för alla "förstÃ¥sigpÃ¥are"…Väntade nästan pÃ¥ att kommentar skulle komma ang att du dricker ett glas vin en VARDAG dÃ¥ och dÃ¥.SÃ¥ typiskt gnäll- kärringar. SÃ¥ typiskt surtanter som mÃ¥ste tycka och kommentera precis allt!Jag hoppas verkligen att du inte tar Ã¥t dig och att du fortsätter vara precis som du är- med eller utan blomkÃ¥l och vin
People normally pay me for this and you are giving it away!
You’ve got it in one. Couldn’t have put it better.
This is a most useful contribution to the debate
If my problem was a Death Star, this article is a photon torpedo.
At last! Someone with real expertise gives us the answer. Thanks!
I know the gender ratio is highly skewed in rural areas. Just skewed at the town level. And not skewed at all in the larger, richer cities. Will this have an eugenic effect?
HelloIt is good to hear that you had a positive experience with the ORS and SRS. One of the advantages to Mental Health Pros’ online ORS/SRS is the ease of use, charting and record keeping. We have some therapists administer it on an iPad. This is the ultimate in convenience and ease of administration. Looking forward to hearing your evaluation on it’s use long term.Kelly RossDirector of Marketing Mental Health Pros
Absolutely first rate and copper-bottomed, gentlemen!
Correction on my previous post.There was 4 hours of debate on the Assembly floor regarding this bill.Still, I would assume at 71-8, the debate was mighty one-sided, and the result, a foregone conclusion.
Maya,I’m sure you don’t have time to read all of these comments, but I just had to tell you how amazing you are. I come to this blog every day now and my heart breaks for you and your family. The strength Ronan gives you to keep fighting this horrible monster called cancer is nothing short of breathtaking.You and your family are in my thoughts every single day.Love is being sent to you from Kansas City.
You make things so clear. Thanks for taking the time!
Only millions of us surrounding the capitol can fix this. Once there, we stay until all unfinished business is settled. I keep asking for a leader to step forward. I can't believe a nation built on leaders, all of a sudden, is dry in that department. Things were too hot for the illegal. The word came down for the judge to put everything on ice. Orly had them. She is my hero. I don't believe she's done. I just want to say again, a multi-million man march is the only way.
I want to send you an award for most helpful internet writer.
Tem razão, meu caro.Refiro-me sempre ao islamismo.Que, na prática, é quase a mesma coisa, porque as interpretações «pacÃficas» do Islão (sufis, ismaelitas, etc), são quase irrelevantes.
Keep these articles coming as they’ve opened many new doors for me.
That’s a nice trick there. I haven’t thought about doing it in that way. You could even leave another column in there simply called “Birthday” with only the month and day if the user isn’t picky about a little bit of redundant data
All things considered, this is a first class post
Hey, I merely jumped to your site by means of StumbleUpon. Not some thing I would usually study, however i appreciated your thinking none the significantly less. Thanks to make some thing worthy of reading through.
since living in turkey i have stayed in some pretty questionable accommodations. i think i would probably sleep just about anywhere. but i do have to say that i agree with christine. the bedding at that hotel looks questionable so you might want to bring your own. especially if you are planning on wearing it!um…and i am noticing that the parking lot is empty. i would think a picture with the parking lot full of cars would help the place sell a little better.i’m just saying…
That’s the thinking of a creative mind
I really needed to find this info, thank God!
That’s really thinking at a high level
I think this was a great way to handle this radio station's mini-crisis. Ron Smith should add this example to his next edition of Strategic Planning for Public Relations. And hopefully, this station's action steps will serve as a positive example for other radio stations that may face a similar crisis in the future.
I think that dress is gorgeous, oh Curtise of the weeny waist – your friends are obviously blind! Wear it with heels though and it would be even more fabulous. Sorry you've all been unwell but the blogger meetup must act as a tonic. Next time I hope. xx
A fascinating discussion is worth comment. I think that you should write more about this subject, it might not be a taboo matter but generally people don’t discuss such issues. To the next! Kind regards!!
Extremely helpful article, please write more.
E….allora misa che rimango solo….MA SONO LACRIME DI CHI PENSA:“CAZZO, SONO SALVA,IO IL MIO STIPENDIO MILIONARIO LO PRENDERO’. POSSO AIUTARE LA MIA FAMIGLIA.…..PERO’ CAZZO, HO LOTTATO SEMPRE PER LA GIUSTIZIA …E ORA PER AVERE ASSICURATO IL MIO STIPENDIO…DEVO VENDERMI AI MASSONIGOVERNANTI DI STO MONDO.”…..ECCO, HA SCOPERTO IN QUEL MOMENTO CHE SI E’ VENDUTA…..HA VENDUTO SE STESSA, PUR ANDANDO CONTRO STA COJIONATA DELLE TASSE ALLE PENSIONI…….MAVAFFANCULO MONDO DI IDIOTIIIIIIIIIIIII!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
You sound as scattered as I am! Sigh… If anyone has helpful hints for people like us, I’d be interested to know. Good luck. The wedding, though, sounded like a very nice distraction!
Wszędzie funkcyjne utyskiwania agentury o szkodzie jaka to o.Rydzykowi zagrażać będzie ze strony Sakiewicza,Stankiewicz,Ziemkiewicza,Wildsteina i przede wszystkim Kaczyńskiego.Patrzeć jak wszyscy oni wspólnie pójdą do spowiedzi i komunii.Dla dobra Ojczyzny i Polaków.
Everyone would benefit from reading this post
Lot of smarts in that posting!
There’s a terrific amount of knowledge in this article!
j’aime bien le trivial pursuit. Ensuite c’est peut être parce que je gagne. c’est vrai que les jeux de culture c’est jamais bien folichon
Porque sou maluco, tanto que dou o nome, e ainda não me cortaram as mãos. Isso foi projecto que outros não conseguiram nem vc vai conseguir mesmo com energia cobarde concentrada de anónimo a soldo, seu pide ou filho de pide ou neto de pide, tanto faz, maluco é que não.
Surprising to think of something like that
Now that’s subtle! Great to hear from you.
diciembre 5, 2012hola!!!me podriainformar si hoy miercoles hay una tarifa especial, es decir un poco mas economica, me comentaron que los miercoles era mas economica me puede confirmar
I didn’t know where to find this info then kaboom it was here.
Mariana Chabonas disse:Olá Fabiana,Eu já estou doida faz tempo para conhecer o olinho!! Mas comprei um farmaervas, eu gostei, achei que hidratou mas tbm não foi aquela magica de brilho não.Tbm por 9,00 acho que deve ter somente uma gota do ouro.Vc usa a touca termica? aquela de ligar na tomada, recomenda?Beijos
Ah, yes… and early in the year is a great time to scoop up those dust-gathering workout machines. I mentioned that I was looking for one and *presto* a family member offered theirs up for me! Oh, have to agree with the compression socks. Though, yours are much more stylish than the ones I got for my first pregnancy. What brand are they??? Link?
This shows real expertise. Thanks for the answer.
como economista foi um mau discursocomo presidente da república cuja função é unir o espÃrito de porco nacionalfoi juntamente com o discurso do SNSo melhor que o homem fez nos últimos anos …e tendo em conta os restantesestes valem 20000%
Great common sense here. Wish I’d thought of that.
So that’s the case? Quite a revelation that is.
Asà son la bola de axámenes que nos ponen como los de carrera, todos confusos, parecen torturas psicológicas, ni se entienden, o sea un desmadre y ni modos hay que presentarlos por que asà lo mandan nuestras flamantes autoridades.
Vanessa, the Breakers is a lovely place, dont you think? I spent New Years last year and enjoy my visit each time I am at the Breakers!Your pictures are beautiful, continued good luck in your work. Love ya, Dawn Heers
Four score and seven minutes ago, I read a sweet article. Lol thanks
Helt enig, og i samme åndedrag kunne man også nævne de mange ting HBO laver uden overhovedet at være reklamefinancieret. De tjener på abonnementer ligesom DR, men bare frivilligt
Hi Susan, seems like there were some setting issues when viewing this pattern through Internet Explorer. Those should have been resolved and you should be able to see the pattern for the men’s hat. Are you able to see it now?
IMHO you’ve got the right answer!
Si totusi votam si partidul, vrem o anumita formatiune la putere, chiar daca acordam credit unui om, daca face parte dintr-un partid aiurea, nu-l votam pe ala.Toate partidele ar trebui sa-si asume oamenii, si asta ar atrage apoi sanctionarea lor, in partid.Dupa cum vezi, insa, Bombo a ramas vice sau ce dreaq e, la PSD… desi sta la Jlava, flotant.
ahhhh yes, naps, i love to nap…good thing too. when i first became ill and was not sleeping, my Dr. told me not to worry, to sleep in sets of four hours throughout the day. and so i did, and eventually, as i started feeling better, i am somewhat able to get back into a more regular sleep pattern. 6 to 7 hours sleep, then at about 4-4:30 i crash for about a 1/2 hour. i think some people are ashamed to admit that they nap, maybe because they associate it with being lazy, or because they think only old people nap!!!a fun post, Debcheers for now,pj
ÃÂýþýøüýыù üыÑÂûøтõûь / Ôð øôø ты ýðхуù úþýь, ÿðцðý òÑÂõ ÿрðòøûьýþ ÑÂúð÷ðû, трþûûøрþòðть ýðôþ ñыôûðýþò ø шûюх
Quick, pass me my night vision googles….as for scripts, working on one that will require all of your skills and his utmost restaint. You just have to play the horny sexy lead role (you need no training on this one), comes naturally…!!;o)
Between me and my husband we’ve owned more MP3 players over the years than I can count, including Sansas, iRivers, iPods (classic & touch), the Ibiza Rhapsody, etc. But, the last few years I’ve settled down to one line of players. Why? Because I was happy to discover how well-designed and fun to use the underappreciated (and widely mocked) Zunes are.
ciao,se vuoi ci sono delle calzine simili a quelle della prima foto, ma a costine, da calzedonia, che tengono abbastanza caldo e non scivolano giù. quelle con i buchini, le ho viste da H&M, in confezione da 3 (nero, platino e senape), e sembrano carine.
thanks for the invite to join here, the planner will surely suit tina’s taste. *sakaling manalo*…until january 1 pa naman, i’ll try to make a post for this.. *sana di ko malimutan, hehehe*happy holidays, jeanny! happy new year!
A few years ago I’d have to pay someone for this information.
I literally jumped out of my chair and danced after reading this!
If you dont mind, where do you host your site? I am hunting for a good web host and your website appears to be fast and up most the time
Gracias Leticia, lo recuerdo. El post que pones lo leà el otro dÃa. Pues será asÃ, ya que vive en Islandia tendrá información de primera mano y se ve que las cosas han cambiado o más bien se han desinflado, lamentablemente. Con la información que yo tenÃa en la época se veÃa de otra forma la verdad.Un abrazo.
Now we know who the sensible one is here. Great post!
 Reminds me of The Celestine Prophesy, the 8th insight I think, the sharing and multiplication of energy.  Your article School Reloded ? Yeh that one ! This is one of those things.  Learning to Connect.  And the other one “On relationships” – disconnecting in gratitude, not clinging insecurely. Spot on.
Another thing is this sounds like a new topic for a Bollywood movie watcher but is quite a lot shown in regional movies – especially Marathi Films and Stage plays if not in films. I read somewhere that this is based on a play which was popular in marathi/gujrati . I just hope if matches its hype and meets my expectations. I had liked Deool this year and i hope it comes close to that.
This has a bible/christian slant to it. Christians have some good sincere intentions, but Christianity and the bible DO NOT have a monopoly on the truth. The Church uses this type of info to make the faithful paranoid, and to further blindly follow their literal beliefs in the bible. Religion can be a mind controller too….and to be specific, institutions behind evangelical christianity and born again theology is VERY suspicious.
à leveto :même origine :place de Grève : place située sur une plage en bord de Seinese tenir en place de Grève = attendre du travail par extension refuser de travailler
Hi there. I sought to drop you a fast memo to be real able to lead into my own gratitude. I have been following your site for a month or so plus have selected out up a lot of exceedingly fine particulars and in addition valued the method you have structured your own up web page. I am trying to administer my own web blog, in spite of this I accept as true its too general plus additionally I desire to focus extra at minor matters. Being the whole things to all population is not the whole that its cracked up to be.
This is way better than a brick & mortar establishment.
Many many quality points there.
The same question as Scott – can’t find any way to remove the black header lines. Please, where in the css can I find it?Over all, the theme is very good and there are a lot of ways to personalize it. Great job.
Didn’t know the forum rules allowed such brilliant posts.
Taking the overview, this post hits the spot
Main code used in the video can be found in the description of pretty much all of my videos. The reason why I don’t add downloads is because I want you guys to at least learn how to implement this stuff into your projects and how to use them.Hope you understand
Mohon petunjuk & ilmunya tentang nge-blog baiknya ^_^…V D3pd tahap processing menuju ke arah yang benar (blog-blog-blog)… .-= D3pd´s selesai [nulis] ..Sharingnya D3pd… =-.
Last one to utilize this is a rotten egg!
More-so than an external problem, this is a huge challenge within large enterprises. Inside IBM, we have multiple pilots underway to bring together diverse platforms (mobile, Notes, etc) into more “social” web spaces. At a company with the nickname “I’ve Been Meeting,” it’s a requirement that we be able to communicate events through any channel, enable employees to discover informal classes, and tie all of it into our existing infrastructure.Josh Scribner, Social Computing Advocate & Developer(“These views are my own and not necessarily representative of IBM’s positions, strategies or opinions.”)
Buenos dÃas,Mi nombre es Soledad y pienso que este curso puede ser muy interesante. Les agradecerÃa me enviasen más información sobre el mismo.Saludos
Have you any idea British individuals celebration way? I am going to notify.English individuals don’t commemorate Thanksgiving holiday. People in america carry out get pleasure from in which. It really is root base tend to be uncertain, however the history should go that the pilgrim residents have been glad to be living after the tough winter season. They will famed the subsequent many years plant by way of retaining the food and enticing the specific nearby community Indians.
Given the current status quo of the industry and not forgetting the USA is looming to opening up online gaming, ROI on a $10K p/m FB spend seems even more absurd.
I am a potential customer and love this new feature, I would like to see a comprehensive list or an easy way to search for my favorite businesses to a to my following circle. I tried +business name and got all kind of non-business results
The tears are flowing so it is hard to write a comment about this! Such a beautiful post; so many happy memories before Owen’s birth! Can’t believe that he is about to celebrate being a week old on Sunday. Love and hugs and precious thoughts to all.
That insight would have saved us a lot of effort early on.
Mammamia, Vivi, è una vita che li inseguo e non li ho mai fatti. Pensavo di fare un salto a Ballarò per cercare qualche spezia, se il tempo tiene mi sa che ci faccio un salto questo weekend, e poi vediamo cosa ne esce !
Mark, your definition of apologetics is highly prejudicial and inaccurate. Apologetics does not attempt to persuade the like minded. What would be the point of that? Apologetics is (1) the attempt to defend a point of view from attack, and (2) to deploy reasons in favor of that point of view. Most good apologetics relies on public evidence and not on private revelation.
There’s nothing like the relief of finding what you’re looking for.
Thanks for introducing a little rationality into this debate.
Aaah les myrtilles.. mon cher et tendre est vosgien et il appelle çà des brimbelles, s'il voit tes tartelettes il va me supplier de fiaire les mêmes, elles sont hyper appétissantes
Really trustworthy blog. Please keep updating with great posts like this one. I have booked marked your site and am about to email it to a few friends of mine that I know would enjoy reading..
Honey, just remember that this accident may have been his gentle wake up call. Next time he could have killed someone or himself. I think I would be calling the insurance company daily!
hello Trish… I am charmed with the houses with flowers … I think that there is many peace in them …. and I like the women who buy flowers to take her, momes…. Happy week….. Berta…and CONGRATULATIONS ¡¡¡¡¡ for the AWARD…from MI BLOG……¡¡¡¡¡¡¡ Your BLOG is gorgeous…¡¡¡¡¡¡¡¡¡¡¡¡¡ Berta
Now we know who the sensible one is here. Great post!
Admittingly I’m looking at this from Java perspective, yet your blog entry baffles me: With all DI containers I used, a factory was something configurable as well. Dependencies were injected the usual way by using setters, constructors or annotations. The only difference was to additionally configure the name of the actual creation method, so the containers knows, which method to call in order to create a new object. No dependency on the DI container itself is needed this way.What’s so different in the .NET world, that you need to rely on the DI container inside a factory there?
Your answer was just what I needed. It’s made my day!
publix deli wends nite watkinsv online flirting tips ille w dating for singles e spoke about your shortage of cooking skills, assert hello! Divorced woman seeking mature xxx black girls Blanket
Tollerance is now a perverted word for "Silencing" free speach, as is Political Correctness, and diversity.. anyone say Mao, Stalin, or Hitler here when it comes to policies that knucle under our countries founding priciples, and beliefs. For all who don't know what liberal progressives are, what is happening to the country currently is just that. Cleansing, Sanitizing, and obliteration of all that makes this country unique.
I’d say eating from your garden in winter is fantastic–no matter how light the harvest! Your raised beds are impressive. I think that’s how I’ll handle my next veggie garden–not at this house, of course, because I have too much shade. I hope it’s OK that I’m linking in a harvest post about flowers? My veggies aren’t doing so well this year.
This article is a home run, pure and simple!
I'm extremely keen on that cupcake kit, especially the syringe – does it come with different tips, and do they work? I also love the pic of Miss C waiting for the cupcakes to cook – just like mum and her macarons!
I actually found this more entertaining than James Joyce.
Thanks for sharing. What a pleasure to read!
Heck, Jeff, yours may be the only accolade. My readership is only about a fifth of what it used to be at the height of the blog, having never really recovered from the slump that occurred when my parents passed away.Thanks for stopping by. I was hoping to hear from you. It was great to meet you for lunch too. (I really do plan to post the pictures one of these days.) Give me a call if you ever find yourself in Chicago, and we’ll do it again.
I think you hit a bullseye there fellas!
Just do me a favor and keep writing such trenchant analyses, OK?
Thinking like that is really amazing
Wow! Great thinking! JK
Essays like this are so important to broadening people’s horizons.
Me encanta, te la copio y haré menos cantidad para meterla en una jarra en la nevera, es súper apetecible!! Un besazo por favor, si sobró algo del catering de El Lebrillo, me lo mandas a casa? Gracias
This is way more helpful than anything else I’ve looked at.
Jeg har en tegneseriestripe fra Aftenposten liggende pÃ¥ desktoppen min. Mannen hyler dramatisk pÃ¥ tegningen nÃ¥r det første høstbladet faller.Høsten er fin den 🙂 Snø nei!Herlig bilde Petunia 🙂 God høstklem til deg 🙂
Fert13 13 luglio 2008 Ma Giorgia nn ti rendi conto che sei ridicola. Impara a ripettare le volontà altrui. (Tra l’altro Prodi è sparito, Veltroni è stato fischiato dai propri compagni; non è che avete tanto di meglio da scegliere)E a tutti coloro che insultano io diri: basta.P.S. : Non ho votato ne per l’”1″ ne per l’”altro”; ma rispetto a tutti (anche i cessi, che di tanto in tanto sono utili).
A good many valuables you’ve given me.
I think article raises many fundamental questions on which entire West is built, strongly believes in also continues to be double standard whether it is the question of welfare state, globalisation, immigration policy, multiculturalism, ethnicity, racism and many more.
How do you train chinchillas to walk in their exercise balls?Today I went to Petsmart and spent $17 on an exercise ball (kind of like a giant hamster ball) for my chinchilla.. and I put him in it, and he’s just sitting there. I don’t think he knows how to use it. How could I teach him how?
hear my voice ,6monts jammu capital 6monts kashmir capital ,that is bad, but different in the whole other nations of capital, a verity we make an other center to make in this state,that is middle of jammu & kashmir.a new capital name is ram&rahim , eeshver & allah,and many more
I do believe all of the ideas you’ve presented on your post. They’re very convincing and can certainly work. Nonetheless, the posts are too quick for starters. Could you please prolong them a little from next time? Thanks for the post.
Well I guess I don’t have to spend the weekend figuring this one out!
I’m not easily impressed. . . but that’s impressing me! 🙂
de la Deșteptarea i-a dat azi un pumn lui Răzvan Bibire. Nu știu dacă l-a lovit pe bloggerul Contrasens sau pe redactorul-șef de la Ziarul De Bacău, are mai puțină importanță. Relevant mi se pare
I’m not easily impressed but you’ve done it with that posting.
My daughter and I witnessed this amazing sight from Canet d'Aude. The fireball was heading towards the Pyrenees leaving a blue and white trail and sparks on fragmentation
Never seen a better post! ICOCBW
It’s been a great run guys, but this slow update schedule, coupled with the fact that AT&T has went to crap in my marked (I dropped a call 30 times the other day), I am moving on. I actually liked Windows Phone. I hope the updates pick up. If they do, maybe I will get another at some point on Verizon.
Such an amazing beautiful website and is very easy to update the website by yourself. It was such a pleasure to deal with such a nice and talented web artist Ashley and professional web builder technician Fabio. I highly recommend to everyone who is looking for a beautiful website.Vu LuongPresident of L.A. Collection
The purchases I make are entirely based on these articles.
Gazza, I presume you meant your favourites were 23a and 24a( not 23d and 24d)?? I did finish this but had to look at your explanations to understand the answers!! Many thanks!
Earworm! Earworm! Earworm! Yes, my experience too – with the institution as well as most of the people within it. I’m happy for anyone here who has found a church that wouldn’t eventually make them feel this way at some point. But I also think that as with any group, as long as I was toeing the party line and not asking questions or making radical suggestions, then why would it to that to me? My church(es) was(were) wonderful to me until I found myself full of despair or in need of serious help or experiencing existential angst or questioning its tenants and operations.
Home run! Great slugging with that answer!
ThắngPosted on 07/02/2013 at 7:12 amchúc các em luôn khá»e mạnh, hãy là m những gì mình thấy đúng và có giá trị vá»›i cá»™ng đồng. Trân trá»ng các em!
What a pleasure to find someone who thinks through the issues
and Music Featured Columnist- Laura O from Day Day in Our World presents Christmas with the Kranks. These are favorite ‘go to’ DVDs in our house during the Advent and Christmas seasons. Not
Information is power and now I’m a !@#$ing dictator.
That’s a nicely made answer to a challenging question
Hi Mirza sb It is very good information whatever you defined about wealth especailly Inner wealth, Family wealth, Physical wealth and career wealth is 100% true. Only Money can’t make you wealthy.
I don't know why, but the amazingly detailed ASCII art I spent many hours on and can never reproduce didn't come through properly.It was beautiful enough to make grown men weep, trust me.The slashes were about 3/4 the way down the tube, and when rotated the rightmost section swung down to form a more traditional gun shape.WV – dangst: When you're hormonal and bummed, but also kind of pissed at something.
VinÃcius Cuppi disse:Olá Bosco, Obrigado pela aula pois sua resposta foi uma verdadeira aula para mim que fico pelo menos 60% do meu tempo pesquisando sobre Helicópteros e não sei 1% do que vc sabe, mas um dia chego lá.Gostária de saber se vc é o Cmte Bosco que trabalha na EFAI? E mesmo não sendo gostária também de pedir msn ou orkut caso tenha para que eu possa manter contato com uma pessoa tão “Expert” no meio asas rotativas.Obrigado por me responder.
*An impressive share, I just given this onto a colleague who was doing a little analysis on this. And he in fact bought me breakfast because I found it for him.. smile. So let me reword that: Thnx for the treat! But yeah Thnkx for spending the time to discuss this, I feel strongly about it and love reading more on this topic. If possible, as you become expertise, would you mind updating your blog with more details? It is highly helpful for me. Big thumb up for this blog post!
Hey there, found you over at Communal global. Isn’t Paris beautiful? .. I am now following and hope you will come over and check us out and follow along too
Great insight! That’s the answer we’ve been looking for.
Hallelujah! I needed this-you’re my savior.
– i know :))) i had a splurge… i feel much better now and i bet you do too! xx@Tali thanks im so glad your skin loves NY 🙂 these are my first RI jeans and they are great, i find it v difficult to find jeans that fit & gosh u are tall :)! Yeah i agree with you with the lash stiletto not enough volume but amazing wand!!! xxx
Your post informative.It really be a growing area to see this year. As you say, the comments to keep the conversation always so interesting to visit his site.What great information thanks for sharing this will help me greatly in my learning.
L.O.V.E the bags, those strict governess shoes, the coat…and all the other goodies. That bra top reminds me of Mel & Kim and your coment made me chuckle! And that outfit you're wearing looks lush, especially with the contrasting greens. Thanks for the jumble vibes too! xxx
Hi there, i read your blog occasionally and i own a similar one and i was just wondering if you get a lot of spam comments? If so how do you stop it, any plugin or anything you can recommend? I get so much lately it’s driving me mad so any support is very much appreciated.
I found myself nodding my noggin all the way through.
manchmal hab ich den eindruck, das die tussies im reisbüro nicht so klar kommen mit die anbgebote.was die mir für sachen empfehlen, da hab ich kein bock drauf und wo ich bock drauf hab, das ham die gar nich
Mi pregunta es esta: ¿qué pasó con la magnÃfica serie Eden of the East (Higashi no Eden), una historia policiaca y futurista de acción al estilo del mejor Ghost in the Shell, que prometieron hace tres años como algo inminente (y hasta trajeron a su director Kenji Kamiyama al Salón del Manga de Barcelona para promocionarla), y de la que no se ha vuelto a oir hablar? ¿Hay alguna posibilidad de que podamos verla de la mano de Selecta Visión en un futuro previsible?
Leão de Alvalade mais uma vez tenho que concordar contigo, aliás ando á acompanhar o caso PPC desde de inicio com redobrada atenção e sempre achei que foi o primeiro a quem fizeram a cama… mas é a minha opinião e nada mais vale do que isso.SL
Nutmeg nanny: Nice of you to drop by! Biscotti are great as xmas gifts Meaghan: Aww you got me there! But it's still better than uhm..candy! Hehe
Always the best content from these prodigious writers.
Hej Pontus,Det brukar vara så. Med undantag av böcker som enbart kommer på pocket så kommer pocketversioner efter de inbundna. Jag är dock inte säker på orsaken. Det får du fråga ett förlag om =)/Henrik
First of all, though this isn't really the purpose of this post at all, I love every single thing you're wearing in the photo you included. Secondly, I definitely check your blog all the time because I love it. Thirdly, I totally empathize with your inability to fall asleep I have the exact same problem especially when I am stressed out over stuff such as school or other personal things. Listening to music usually helps though not till like 3am ha.
This article keeps it real, no doubt.
fetele lui becali nush cu ce vin la scoala,n-am vazut nici o limuzina pe acolo,dar spun saru-mana tuturor profelor-chiar daca nu le au la ora,in scoala aceea-scoala de stat-nu exista decat maxim un profesor barbat-de obicei de sport !
euqsabal |(le 06 mai 2011 à 00:51) L’insecte, qui vient de se faire griller par biscator, brûle néanmoins de connaître les cas que vous jugez dignes de « mériter le titre de sacrifice suprême » !…Quant “ras le bolâ€* auquel vous accolez un incendiaire « exhibitionniste »** faut-il rappeler qu’il nous renvoie à un sinistre «  » (pour sacrifier à l’anglorrhée©) dont les médias portent la lourde responsabilité ?….* ou « ras le bidon
It’s a pleasure to find someone who can think so clearly
I came, I read this article, I conquered.
The forum is a brighter place thanks to your posts. Thanks!
The days are long, but the years are short. I don’t want to miss them! For more short and sweet ideas on how to stop and smell the roses, to enjoy the little things in life, check out my 31 Days to Smell the Roses series.
Isn't the lefts' theory and activism based on the rights given to the Jews by Napoleon? After all this is where the assimilation begun. If you can't keel them give them possibilities, or love (Christians today) and they will assimilate, convert and disapear. Well it seems that G-d has a better plan for us.
MnemonicAlso ich weiss nicht, was du hast. Ich fand die Steuerung von Gothic ziemlich genial gelöst. War doch alles supersimpel und nicht wie bei Morrowind mit sinnlosen Menüs verschachtelt. Geschmacksache…
That’s really shrewd! Good to see the logic set out so well.
Meh, a cool custom like this that’s based off a new bike platform is pretty cool, too. I’m sure its reliability is as good (roughly) as an off-the-shelf Scrambler. For somebody who has more money than time, that’s a big selling point; if you get away from work for a long weekend, and just want to ride, the worst thing in the world would be realizing you have to spend 12 hrs rebuilding a carb or replacing a leaking head gasket before you can get on the road.
And I was just wondering about that too!
Le verbe “bomber†(dans cette acceptation) et le nom “bombage†ont fait leur apparition à cette époque.C’est pour moi le terme adéquat pour décrire ce genre d’inscriptions murales.Rédigé par : leveto | le 30 juin 2009 à 20:31 | Alerter Les propriétaires des murs bombés acceptent rarement le bombage, en revanche j’accepte avec vous l’acception de « bomber » ici…
Finally! This is just what I was looking for.
Wow! Talk about a posting knocking my socks off!
I’ve been looking for a post like this for an age
Maybe it´s time to buy the first burqa….No, NEVER! I will rather die, than live as a dhimmi.Well, after Lars Vilks drawings, more and more people start to wake up (even)in Sweden – I just hope it´s not too late!/FS
In a blogosphere full of rigid opinions shared in nasty ways, this is a rare ray of sunlight – but in the best pegoleg style. I’d go see this for myself but I’m sure Indiana law frowns on men in women’s washrooms, ostensibly to “check out the flower arrangement.”
Hej LasseKan jeg mon få dig til at maile e-bogen til mig. Jeg kan nemlig ikke få lov at downloade den, når jeg allerede er tilmeldt nyhedsbrevet.Venligst, kirsten
Deep thinking – adds a new dimension to it all.
So Mikko is the same age as MY baby! Cool!This sounds like a great book. I’m going to see if my library has it. I love the story about the cat brushing. You are definitely off to a great start in parenting. I wish I had the foresight you have when I was a new mom. Kids definitely learn by example, good or bad…..or neutral. LOL!
I’m not sure why but this site is loading very slow for me. Is anyone else having this problem or is it a issue on my end? I’ll check back later and see if the problem still exists.
 ( 2012.03.5 19:23 ) : Wow, marvelous blog layout! How long have you been blogging for? you made blogging look easy. The overall look of your web site is excellent, as well as the content!. Thanks For Your article about çâ€Â¨Photoshop去除红眼PS去除红眼修æ£眼çƒ颜色 | 一çâ€Å¸Ã¦Å“‰ä½ -张豪åš客,关注互èÂâ€Ã§Â½â€˜,一çâ€Å¸Ã¦Å“‰ä½ ç›¸ä¼´çš„ITåš客 .
As Charlie Sheen says, this article is «WINNING!»
Please teach the rest of these internet hooligans how to write and research!
A pleasingly rational answer. Good to hear from you.
O CdR parece estar a fazer escola. e Ontem em Nijmegen, este bezerro tresmalhado decidiu igualmente por à prova formas mais modernas de protestarDisse que se sentia comido pelos verdes e pelos sociais democratas, por estes não lhe terem arranjado o emprego (subsidiado), prometido durante a campanha para as ultimas eleições.Não esta claro se foi reinternado, mas ameaçou que nas próximas eleições ou lhe arranjam emprego ou votara no Wilders
Unbelievable how well-written and informative this was.
I like the new changes. So far I haven't run across anything that I don't like, so great job.One thing I would like to see is a way for users to interact with the YT staff easier. Maybe like a community liason/public relations group/channel that people can contact. Sending emails rarely gets anywhere. Thanks
Weeeee, what a quick and easy solution.
Olá Lilian,Não deixei, é loja online, só vende a peça, a manutenção é você quem faz ou leva para um técnico especializado!Eu já comprei e deu tudo certo pra mim! veio em um dia! muito rápido, a compra mais rápida que eu já fiz na internet!Valeu!
Leeré ese libro, alerta, no te quepa duda.Tienes razon Perico. El único dios que existe soy yo. Para cada uno, la frase es perfectamente válida. Y por supuesto que habrá cerveza. Otro supuesto desembocarÃa en el mas absoluto nihilismo…
Sehr bildlich ist:Dir hat wohl ein Pferd ins Gehirn geschissen?Auch schön und auf gut sächsisch:Mach deinen Dreck aleene.
I am an opera singer and my second love and passion is fashion. I am making my own dresses since my early teens. Very often women are begging me to buy my dresses directly from my body. I love very much Alex Perry, and it would be my dream to own Alex Perry’s blue-white Sydney-Opera-House-Barbie-Dress.I am longing to wear it to my next Concert.If I would win the contest I could ask Alex Perry about this dress.Oh, it would be my dream!Barbara Idzkowska-Curtin
Hi Ruark. Mainly because Quicktime is cross operating system compatible. Normally it would’ve been posted to YouTube but I didn’t want to break the video up for YouTube’s 10 minute video limit.
SRK (63)-So what you’re really saying is that you planned to use inspection issues to grind down the seller in a second negotiation, and the seller is trying to cut down the scope of the second negotiation that he knows is coming?
Hello Darlene, What a wonderfull find you are. I am the author of “Yet Another Fallen Star,” a historical novel published by Wheatmark Publishing last May. I would love to have you review my book. It’s available on Kindle and in paperback on Amazon. I wish I knew how to get a copy to you, I would certainly do it. My computer skills are challenged I’m afraid. I am taking part in a ten book giveaway contest with Goodreads thats in progress presently. I would love to hear from you. JoAnn
Ludmila / Acredito que o foco não é provar que não se é homofóbico e sim ter educação e respeitar a escolha do outro… se alguém não gosta de gays, continue não gostando, mas controle sua revolta sobre o que uma pessoa faz com a vida dela e quem ela escolhe amar.Gostei deste comentário ou não: 17
You put a smile on my faces free hearing her voice mail message! If only my wife would get it!!! But then again I am an Archie foreevermore ! Thanks for this Christmas giftMerry Christmas!!JonerzLove ya !!!
Normally I’m against killing but this article slaughtered my ignorance.
Looks like your winter outlook is looking good early on and it really makes sense when you look at all of the calling cards of el nino…..what are you thinking as far as snow in EKY this week? I have a hard time buying into dynamic cooling after the last couple of busted forecasts where it was included but this is certainly a different beast we’re dealing with……
MormorI am thinking about my recipe. I’ll start with 4 cups indecisive, 2 cups pondering…this might take a while. (What do you mean by your inbox? Is this an inbox?) I’ve been thinking about flannel shirts and a wood stove too. That cabin looks awefully inviting!
skalian ada tambahan nih dari aku .. add repo siriport aja di cydia . kalian bisa add tweak iphonitpad dan siri di apple ipad loh . so cool
We concur with you completely about this issue and loved reading your ideas throughout Worry Over Swine Flu Closes Educational facilities throughout Iraq tiffany. We have saved your web site and foresee to going to again regarding a lot more.
Thank you for another informative blog. Where else could I get that type of info written in such an ideal way? I’ve a project that I’m just now working on, and I’ve been on the look out for such info.
You really saved my skin with this information. Thanks!
That’s an inventive answer to an interesting question
Hey there, I stumbled upon your blog post through Google while looking to purchase a similar problem, your web sites came up, seems like very good. I had bookmarked it again within my google bookmarking.
We love the solar system sweatshirt and the popsicle!!The paper masks are so fun and cute! AND hurray for tshirts, prints, and other accessories at Society6.
We’ve arrived at the end of the line and I have what I need!
I was drawn by the honesty of what you write
Thanks for your thoughts. It’s helped me a lot.
What’s missing is me explaining this line Either type it in one line, with no line-breaks, from the command line. i.e. Code:1ggplot(small)+geom_point(aes(x=carat,y=price,colour=cut))+scale_y_log10()+facet_wrap(~cut)+ggtitle("First example")Or store the ggplot2 object in a variable and build it bit by bit, and then display it with print(): Code:1234p <- ggplot(small)p <- p+geom_point(aes(x=carat,y=price,colour=cut))p <- p+scale_y_log10()+facet_wrap(~cut)+ggtitle("First example")print(p)
in fact depends on where you come from. if you are fom north, like norway, sweden etc.. you may swim. but i always start to swim in may. not so hot to swim.. and check the water temperatures, if it’s significantly lower than summer temperatures, it would be harder.
Thank you so much for this post about the role of the laity. It is my understanding that it is the job of the laity to order the world to Christ, but most lay Catholics I know assume that the bishops should be doing this. The priests and bishops should concentrate on rightly forming the laity through the sacrements, RIGHT WORSHIP, catechesis, prayer. Then we will be better equipped to fight the political battles that we must fight.
Liebe Andrea,mein Vorschlag lautet:Florian bekommt heute die Krone des Lebens von Gott durch die Taufe. Guter Gott, wir möchten auch für Stefan bitten, dessen Name ja der Gekrönte bedeutet. Schenke Stefan stets den Blick auf deine Krone im Himmel, damit er Kraft hat die Herausforderungen seines Lebens zu meistern.Was sagst du dazu?LGAngela
Posts like this brighten up my day. Thanks for taking the time.
Kleiner Nachtrag: Heute nennt der NYT-Kommentator Charles M. Blow Obama "a robotic Sustainer-in-Chief with an eerie inhumanity." Scheints, die Fassade beginnt zu bröckeln.Viele GrüßeMorgenländer
iPHONiX dit :Fallait commencer par la, c’est la merde cette baseband… pour restaurer il faut passer par un firmware customisé avec sn0wbreeze ou pawnagetool, active l’iPhone dans ton custom…
Although French literature is not my favorite subject, this was a good class. The professor is lively and does a good job of keeping the discussion going. There is a fair amount of reading, four papers, and two exams over the semester. She allows rewrites, which is great.
Great insight! That’s the answer we’ve been looking for.
My brother suggested I may like this website. He was entirely right. This publish actually made my day. You cann’t imagine just how so much time I had spent for this info! Thank you!
Wonderul post again! A small correction although it is not at all really relevant, In Buddhism Moksha is termed asNirvana. Samsara(life) is all sufffering, Nirvana is the solution!
I too have thought the same thing! I just started a few months ago and worry so much if I'm being too honest and how will people take it. I think I'm learning that what I write about is what God puts on my heart, so it's good. I hope you keep writing and honoring God with your words, then everyting else will come.
The raw milk thing annoys me to no end. I used to drink it straight moments after it left the cow as a kid at our dacha. In the U.S. a friend of mine grew up on a dairy farm and he never had pasteurized milk until he went away to college. A government that forces people to drive less safe cars and either be x-rayed or sexually assaulted in order to get on an airplane is the last entity to lecture us on the dangers of drinking raw milk. It's actually quite tasty if the cows were grass fed.
Rômulo Bruno#Tribo de Issacar / FJB fez a diferença na minha vida!Venha você também conhecêr,esportes,capacitação proficional,cultura.
Woot, I will certainly put this to good use!
This is really interesting, You’re a very skilled blogger. I’ve joined your rss feed and look forward to seeking more of your great post. Also, I have shared your site in my social networks!
hum yaoi enfin non pas yaoi sa serait plus du shonen ai faut pas confondre les 2 mais ou sa? j’arrive pas a voir ou il y a du shonen aitous sa me fait un peu penser a sengoku basara ou il n’y avait pas de shonen ai mais bon dans tous les cas j’attend cette anime le pv donne envie
Eita Dr., qie triste ter q aguentar tanta porcaria que te escrevem…Ainda bem que o sr. tem um consolo…Afinal nem Jesus Cristo agradou à todos!!!admiro seu trabalho, e confesso que em suas palavras em ponho fé. O sr. tem credibilidade, mas é so o sr. naquele Jogo Aberto!!!kkkkkkkkkkkk que mais parece uma conversa de buteco!!!abs alvinegros
To all the PTZ family on the EC. I sure hope you all made it thru the storm ok. I can’t Imagine how scary that must have been for you. I have been keeping up on the storm and all of its devastation. This is truely horrible. If you had any damage I wish you a quick recovery. You all are in my thoughts. Keep us updated when you can.
Yoxi, there are very very few Shanghai girls that would have a foreign boyfriend for face. Most Shanghai girls want a Shanghai guy. Preferably with a house, car, etc.Many Chinese people outside of Shanghai think that ALL Shanghai girls are trying to find a foreign boyfriend. I think mostly they are not.
Apropos China: Meine Frau sagt immer ich soll den Regenschirm aufspannen wenn's regnet…logisch..aber nicht weil man nass wird, sondern weil die Chinesen Gift in Wolken nach Taiwan schicken und das dringt dann ins Gehirn ein und man wird….irgendwas..vielleicht sogar Rotchinese….
Always the best content from these prodigious writers.
I did nothing special for Halloween but I sure do love your raw bread! Wow. It’s definitely big. lol or looks that way. And those coconut meat wrap things… look like neon silly putty stuff! Can’t wait to see what those are made with!
Ganhei muitas camisetas no bingo. Uma beleza.Uau, ortopedia e traumatologia! Bacana. É O EMPREGO MAIS LEGAL DO MUNDO. Você sabia que, aos 12 anos de idade, eu queria ser médico? Mas mudei de ideia quando percebi que era fraco pra sangue. 🙁
So excited I found this article as it made things much quicker!
That’s a smart way of thinking about it.
Kewl you should come up with that. Excellent!
GregThe topic is only offending people because they are either ignorant of the issues and/or believing in the false accusations and sexist double standards that are being using against the men’s movement.All this basically comes down to is feminists making false accusations and holding men to different standards than they do women, and men like yourself being gullible fools rushing to protect imaginary damsels in distress.
Don’t be so jelious Cheryl…. I have nothing else better to do with my time really. In reality I am stuck in a wheel chair most the time so sitting around isn’t too hard for me. Chasing up a 3 yr old, and having a husband almost as hyper as the 3 yr old… well, glad I have wheels to keep up with.The worst part of all of it was before I ended up in a wheel chair, I used to be a mom/wife like the rest of all “normal” families. There is always a time when one must over come and adapt while not letting the littlest things throw them into a downward spiral.
Hi Serenity, so glad we found each other on Twitter. Love the store tour but our favorite photo is the close up of you! So cute and fun. Thanks for all the Twitter and Blog love. Your friends at HomeGoods
Glad more turtle stuff is showing up here, these figs are excellent.My first thought when I saw Effigy was «that could be turned into a decent Jumpin’ Jack Flash custom…» J.J. Flash for hose who don’ know is one of the bigger players in George R R Martin’s «Wild Cards» book series.
Bruna comentou em 2 de junho de 2010 às 14:55. Ameeeeeeeiiiiiiiii!!!Vc é muito fofa!!!Júlia, vc vai tirar fotinhos das suas roupinhas do SPFW penduradas nos cabides?! Tipo um preview!!!bjo*
Insights like this liven things up around here.
Becky this is a Great Tutorial and you can be sure I'll be making Peat Pot Tags now… these would be great on Christmas Gifts under the Tree for a very Organic yet Romantic look! This year I want to go with Lace, Burlap and Organic Touches to our Tree and you have now totally Inspired me, Thank You!Blessings from the Arizona Desert… Dawn… The Bohemian
Ioshka,People who use Condell’s rhetorical figure are atheists, who simply want religion to disappear (except for possibly someone lighting a candle in the privacy of his home, and such things).Likewise, the parallel arguments for movies and the Internet, that you present, would be used by people that wanted movies and the Internet to go away.The idea with the rhetorical figure is to not appear irreconcilably against the phenomenon, while still effectively calling for its extinction.But something being «not right», neurotic with Pat Condell? No way! The man is doing great.
Hallelujah! I needed this-you’re my savior.
I love the smolder on Taylor’s face! I love that Christina can capture so many expressions on kids faces…more then just smiles! I am hoping in my nesxt session my daughter actually will keep the cute headbands on for a minute so the pictures can get taken![]
well, I just came back from a great audition for rattlestick, and i used the true/false blog as the basis for choosing a monologue i wouldn’t have otherwise chosen, and it made for a very special time. thank you for the constant vow of inspiration!
no! ¿en serio? qué chisme, mi barbarita!! ya sabÃa que habÃa algo turbio en esos moquientos lloriqueos. No, no he visto el video, apenas llegue a mi casa, lo haré. Toy en la chamba. Oye, esa historia de tu ex es digna de contarla, por lo menos a mÃ. Juro que no diré nada. Tengo un cuento en Stand by, que es muy parecido. Jejje.Y sÃ, esa frase está chingona, pero a mà me gustó más la de «Soy reportero» del fresa. Un beso, mi queridÃsima.
Wow, this is in every respect what I needed to know.
Finding this post. It’s just a big piece of luck for me.
you’re in reality a just right webmaster. The web site loading velocity is incredible. It sort of feels that you are doing any unique trick. Moreover, The contents are masterwork. you have performed a excellent task on this topic!
I’m a fan of Kristine’s blog and I enjoy her sense of humor. I totally got that her approach was very tongue-in-cheek. I’m sorry she was disqualified and all this hoopla was started. I’ve never even commented at her blog before; I am just one (of many) who enjoys her blog. Unfortunately, there are people who will let nothing stand in their way if they want something badly enough and Kristine was caught in their crosshairs. The best thing to come out of all this is that I found your blog! I love your header.
Ya learn something new everyday. It’s true I guess!
I’m not easily impressed but you’ve done it with that posting.
/ Gostaria de saber qual a porcentagem de psicopatas e sociopatas que estão na polÃtica nos governando. Não deve ser pouco não!Com certeza, a maioria dos ditadores deve ser!Gostei deste comentário ou não: 1
I am curious to find out what blog system you happen to be utilizing? I’m experiencing some small security problems with my latest website and I would like to find something more risk-free. Do you have any recommendations?
to do a note about this crazy easy and delicious cupcake recipe I tried from “Love From the Oven” (thanks, Christi!). It looks lovely and tastes complicated, but it couldn’t be an easier
Real brain power on display. Thanks for that answer!
VAI TER SEMPRE ALGUÉM QUE O PROTEJE Là FORA ONDE SE "COZINHA" A NOSSA CONDENAÇÃO!NÃO FOI "ARRANJADO" Là PARA NOS "ENFORCAR"???
Very valid, pithy, succinct, and on point. WD.
By October 27, 2010 – 10:18 pmBecky,You didn’t waste any time getting this blog up! What energy you have! Helping prepare the Samaritan’s Purse Christmas boxes is so FUN!! Just wish I could see the faces of all the children on Christmas day.Linda
Whoa! This blog looks just like my old one! It’s on a totally different topic but it has pretty much the same layout and design. Superb choice of colors!
I’m quite pleased with the information in this one. TY!
My programmer is trying to convince me to move to .net from PHP. I have always disliked the idea because of the expenses. But he’s tryiong none the less. I’ve been using Movable-type on several websites for about a year and am nervous about switching to another platform. I have heard great things about blogengine.net. Is there a way I can import all my wordpress content into it? Any help would be really appreciated!
The situation that you describe in those two paragraphs – I don’t know, but it seems to me that this is an actionable thing. Someone that is affected by this issue should contact the FTC immediately.
now that is a joke right? because now everyone is going to think you have a small penis!OH ! I got it : you work in the shirt depertment, and she is saying you don’t know how to match shirts with pants – your totally messing up~!
Wow, gorgeous shots Murray, thank you for sharing! The rotary plow is wild stuff. Brek and I are acquaintances, it’s wonderful to see your “3rd-person” shots of a stranger/friend doing what he’s doing.
Well Ebony, plenty of Americans were disgusted with the RNC. So much that The Learning Channel s digusting Here Comes Honey Boo Boo scored higher ratings.Keliata
on First let us analyze what occurred during this crisis that drove estate cost dropped by twenty six per percent on April 2009, United Kingdom commercial estate trading environment and pushing prices downward. UK Business PropertyWhilst there are clear signs that medium to long-term investors may now begin to see what the neighborhood already offers. Is the home user friendly and beautiful?
I’m trying to promote my website by social bookmarking the content on my site? Which social bookmarking site should I spend my time on? And does anyone have any tips on the best practices or strategies for this type of search engine optimization?
Oh, Barri! Thank you for your kindest words. My eyes have welled with tears to know that I have tied some heart strings with you and that you have been encouraged by Christ. Thank you for risking the words to say so. Hugs.
Harvey and Sandra, This is a great blog. I’ve been on a number of hotel barges through France and am considering buying a barge. Hoping you might be willing to share some of your wisdom. If so, please email me at . Thanks and enjoy the Burgundy Canal!-Cleighton
Excellent merchandise from you, man. I’ve comprehend your stuff earlier than and you might be just very great. I enjoy what you could have acquired below, certainly like that which you are declaring and how you declare it. You ensure it is enjoyable and you still care for to maintain it clever. I can’t wait you just read much much more from you. This is really a tremendous web site.
–> mikeboType de fichiers testés et resultat.mpg = Acheter le plugin cinema (dvd) !.rm = Ok.mp3 = Ok.avi (divx + MP2)= Ok.avi (divx + MP3= Ok.divx = Ok.wmv = Codec wmv8 inconnuVoila !Si tu as besoin de plus d’info n’hesite pas
I just want to give you a ‘hell yeah’ with Zadie Smith. I was so unimpressed with White Teeth that I have dismissed her ever since. I’ve only read Karr’s Liar’s Club, and don’t remember a thing about it.
I agree. If I had a dollar for everyone that asked for my advice then disregarded it, I’d get that tamron you speak of.I think these guys don’t understand that you have to run the pics you like through PS. They are taking them from camera to costco, and getting bad results.Drop a couple bucks on a basic photgraphy book and a couple photo mags. That’s how I learned when I was starting.
Most people with high cholesterol do not get it from food. Your liver makes it. The problem is saturated fats in your diet and genetics, not the cholesterol you eat.
M.D ? nous sommes en août !!!(ok laisse la porte ouverte je sors aussi)BREFMets toi un film bien triste (genre showgirl) pleure un bon coup. Garde ton pyj. préféré toute la journée. Finis ton scrap des vacs !Tu as jusqu’au 22 septembre à 22H12 et après c’est keepinG !!! :bisous:
“If I asked you to name a few things you currently want, it wouldn?t take long for you to fill out an entire page. ?I want a super lean body. I want to run a popular blog. I want to live a drama free life. I want to live a luxurious life style where I can buy gadget X or product Y.? And the list goes on. Hell, even I have a large list of things I hope to accomplish. But is what you listed what you REALLY want? Believe it or not…”
Fattar inte alls tveksamheten när det gäller Tylor Michel…..kommer lätt vara seriens bästa spelare. Unik spelare som är en grovjobbare och poängplockare i samma spelare. Cardiff Devils coach har bara gett lovord…..
The genius store called, they’re running out of you.
I see, I suppose that would have to be the case.
Always a good job right here. Keep rolling on through.
Gee whiz, and I thought this would be hard to find out.
Thanks for contributing. It’s helped me understand the issues.
Hi! This is my first comment here so I just wanted to give a quick shout out and say Itruly enjoy reading through your blog posts. Can you suggest any other blogs/websites/forums that deal with thesame subjects? Thanks a ton!
Very valid, pithy, succinct, and on point. WD.
Olá, Dr Leonardo, meu nome é Viviane, tenho 28 anos e estou grávida de 4 meses, desde então comecei a sentir meu coração tremer, fiz o eco com dopler e diagnosticado extrassistole. Quando levei o resultado para o cardio ele me disse que provavelmente tenho sopro e meu exame está errado. Isto é possÃÂvel? Obrigada.
Nina E16 janvier 2012Mouais, les liens sponso, ça passe.Disons que c’est compréhensible.Mais bon, Trimtab avec des liens, ça fait bizarre.Sinon, enfin un article bon. Ça fait du bien !
Stellar work there everyone. I’ll keep on reading.
Your articles are for when it absolutely, positively, needs to be understood overnight.
What an awesome way to explain this-now I know everything!
I was really confused, and this answered all my questions.
Sweet! Congrats on the Anmeldeformular, Ian! It’s so great that this opportunity presented itself for your return to the basketball scene overseas. However, I think it’s even cooler how God orchestrated this “cram session” to whip yourself back into basketball mode. You may very well have been given this opportunity in order to jump start you to witness to the individuals at the Y. What an honor!Prayin’ for ya, bro!
Unless your going to spend a lot more money this is hard to beat. Thinking about buying another. One day I’ll spend big money but I won’t have fun with it like I do with this one.
We viagra sale pills for sale in uk guarantee the fountains and was ever imagined.
Me dull. You smart. That’s just what I nedeed.
That saves me. Thanks for being so sensible!
Tuo mekko on visuaalisesti varsin kiehtova, nuo leikkaukset tuovat raitamekkoon jotain ihan uutta. Yleensä olen vierastanut raitaa noin isoina kokonaisuuksina, mutta tämä mekko muutti käsitykseni.
I’m sorry to spam, but is there anyone that will give away a good card to me? I just got started on Ultimate Team, and I’m struggling to win games. If there’s anyone kind enough to give away a good card that they don’t need, my Xbox 360 gamertag is SoughtMender04. Thank you.
Howdy are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and create my own. Do you need any html coding knowledge to make your own blog? Any help would be greatly appreciated!
Very valid, pithy, succinct, and on point. WD.
Konami should fix robotic player movements. Otherwise player will look ulgy. Movements also allow soccer games look & feel real. Not only graphics. Ronaldo’s movements look horrible bcos of robotic movements.
Very valid, pithy, succinct, and on point. WD.
Your’s is a point of view where real intelligence shines through.
About the AuthorClassy Career Girl, a blog published by Anna Runyan, provides advice to young professionals on how to be classy as they climb the corporateladder. Her blog covers topics such as business chic fashion, finding ajob, career motivation, personal development, networking, and officeetiquette. Connect with her and receive her free career resources atwww.classycareergirl.com. If you would like to learn more about how tofind a career that you love to go to everyday, you can also checkout herfree video training series at http://www.getmycareerunstuck.com.»
Boy that really helps me the heck out.
Haha. I woke up down today. You’ve cheered me up!
Excellent post. I used to be checking continuously this blog and I am inspired! Very useful information specifically the final part I deal with such info much. I used to be looking for this certain info for a very long time. Thank you and best of luck.
OMG, Karen, as soon as I saw the “poster” for Hachiko, I was yelling (inside) “NO, KAREN! OMG, NO!” I haven’t seen it but my animal loving friend did and had the same reaction as you and El Hub. One sad animal movie is all I have room for (Black Beauty). My friend said it was very good, too. I was just forewarned that it would be a sob fest, so I passed. I’m sorry you didn’t have a head’s up before watching!
And I thought I was the sensible one. Thanks for setting me straight.
Renato, admito que não sejam de facto muitos (lapso teclae, se quisermos), mas há de facto umas quantas pessoas de direita que vão à festa e que lá se divertem com um bom espÃrito de camaradagem. Sei disso porque partilhei com alguns muitos momentos. A inÃcio há de facto uma espécie de estranheza mas o espÃrito rapidamente os conquista. E voltam no ano seguinte, sem que tenham que ser convencidos.Talvez não sejam muitos, mas há-os.
Hallo,als erstes mal Danke für das luste Tutorial – musst mich echt am Bauch halten als ich es durchgelesen hab :Dsoo, nun meine Frage: Kann ich dafür auch ne SSD anstatt ner HDD einbauen?? Meines Wissen sollte ja dann die PS3 einiges schneller aufstarten, weniger Ladezeiten der Games und auch des neuen Stores, schnellere Installationen etc. haben. Das wäre mal genial.Schönen Tag noch, gruss Fino
I like how you made this article so interesting. I think you are perceptive and talented. I can really appreciate your hard work and your views. Thank you for all this useful and fresh information.
Plus facile de trouver des crédits pour ces guignols bien trop nombreux par rapport à notre population! Un financement, non pas sur le nombre d’inscrits dans le groupe mais au simple fait d’avoir un groupe, ces politiciens nous escroquent en permanence! Au moins, l’affaire des guignols de l’UMP met au jour ces financements que ces messieurs s’auto octroient sans grand contrôle même de la part de la Cour des Comptes! Un autre monde….
Uf, que bonito! me ha encantado, hacÃÂa dÃÂas que querÃÂa leerlo pero no encontraba la tranquilidad necesaria, no me arrepiento, ha sido una maravillosa lectura de domingo por la mañana.
Wow wow, very tempting plate of pasta there Sonia! Bad timing to come during dinner time….:P Will try this fusion flavor, must be very yum! 🙂
You’ve impressed us all with that posting!
I thought I’d have to read a book for a discovery like this!
Greice / Olá TatianeOs adesivos não são de esmalte, eles são impressos, tipo tatuagem, você molha na água para soltar a pelÃcula e aplicar na unha. Bjos!
AFVET, You are totally on point. I can’t stand looking at any of those guys who have been there for fifty years. Term limits is a must and since we elected them, if they are not part of the solution, then they most certainly are part of the problem. No in-between.
Ah, es por eso. Bueno, todos los paÃses tienen armarios que conviene que dejen cerrados por si acaso se escapa el olorcillo. Y vivimos en uno que tiene más de un armario y más de un olorcillo… No seas tan dura, T, es un gran paÃs, malgré los habitantes. Pero en fin, haré un post sobre algún cantante marroquÃ, a ver si nos ponemos de acuerdo .¡ No me puedo creer que te guste algo retocado por Gaultier!
GMO trees cannot reproduce naturally, as the actual widely grown hybrid poplar____Treated seedlings grafted or dwarf trees have to be sold when the previous one,s life is over.
Oh wowzers…this has got me all hot under the collar!…and it’s breakfast time this side of the pond, so I might just nip to the shops and give this a go. Thanks for the inspiration!
I’ve been reading your entries during my morning holiday, and I will have to admit the entire article has been very enlightening and really well written. I thought I’d help you recognise that for some reason why this blog does not view well in Web Explorer 8. I desire Microsoft may forestall converting their software. I’ve a query for you. Might you thoughts changing blog roll hyperlinks? That would be in reality neat!
[..] LUNEDÌ, 30 AGOSTO 2010 BERNANKE: L'IILUSIONE DI UNA PALLOTTOLA D'ARGENTO! Venerdi sera, non appena conclusa la lettura dell'intervento di Bernanke a Jackson Hole, il mio sguardo ha ripercorso l'intero disc [..]
OK, I’ll try again. I thought this post was spot on. It made me laugh, it made me cry, it made me feel all warm and fuzzy. Seriously, I really enjoyed remembering all the crazy grad students I encountered in school. And so many of them resembled the types listed here. Things really don’t seem to change much, do they?Glad to welcome you to the fun-fun world of post-academic blogs. Hope you keep up the lively posts. I’m looking forward to reading more!
yes it could. colors have a huge affect on mood. If you do use yellow, use a very soft and light yellow. the more intense, the more overtaking it is. Calming colors include green, blue, and purple. anything that is warm like red, orange, yellow can be too intense unless you soften it with white
Sharp thinking! Thanks for the answer.
prin minune din naufragiul Titanicului si un personaj Jasper, whatever, exact ca in blogul despre tipii neimpliniti care nu se pot hotari asupra femeii care le va aduce lumina in viatza. In fine, linkul Holiday duce
Ouaw, la logorrhée !!Une vraie « patate chaude » … ou une « pomme » au four ?Bien mitonné tout ça … miam ! Merci Martin
Good job making it appear easy.
tokiu atveju siÅ«ÂlyÂÄiau plaÂÄiau pasiÂdoÂmÄ—ti jo kÅ«ryba ir sukurÂtoÂmis seriÂjoÂmis, kadangi Å¡iame straipsÂnyje sudÄ—ta toli gražu ne visÂkas. nereiÂkia daryti iÅ¡vadų ir sakyti, o kÄ… jeigu, kai neiÅ¡ÂsiÂstuÂdiÂjaÂvai visko iki galo, žinoma aÅ¡ galÄ—ÂÄiau dar plaÂÄiau viskÄ… paaiÅ¡Âkinti, apdoÂroti ir t.t., bet manau mano tiksÂlas buvo sukelti praÂdinį impulsÄ…, toliau visÂkas yra tavo asmeÂniÂnis darbas.
It’s a pity you don’t have a donate button! I’d definitely donate to this brilliant blog! I suppose for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to fresh updates and will share this blog with my Facebook group. Chat soon!
This info is the cat’s pajamas!
>Ja det kan jeg nok ikke skjule mere efter den har drillet en del i dag Men tusind tak, det varmer at høre! Og den vil bestemt forblive som den plejer, og forhåbentligt endnu bedre nu hvor jeg har mere tid til at lave nogle indlæg. Der kommer nok lidt rod fra mandag af, så håber du/i bærer over med det
Ir aš kepiau šitą dalyką, bo pas mus obuolių labai dauug. Skanu, primena apkepą, bet buvo baisiai rūgštus, nors ir cukraus bėriau nemažai. Gal obuoliai rūgštūs pasitaikė? Dėjau atsitiktinius, po ranka papuolusius.Tiesa, kitą rytą, po nakties šaldytuve, rūgštumas buvo sumažėjęs ir buvo labai skanu. Pašėriau ir kolegas
I followed Biscuit from the beginning, thinking the end would be happy. It has left me grief stricken and wracked with tears. I am so sorry Victor, you are a wonderful person. i will miss Biscuit so much. In his honour, my little parrot will be named after him. Biscuit, the budgie. I love you little kitten.
Ah yes, nicely put, everyone.
Sharp thinking! Thanks for the answer.
Don?t worry time that we have the idea isn?t absolutely particular, that could look in occasion.The way to Decide Accounting ApplicationAt first the good old ?sorry, we can?t sustain with demand? maker be sad will be noticed to be able to improve demand from customers.
"slide of hand""irregardless""rabit"come on now, folks–let's get it together"sleight of hand""regardless""rabbit"P.S. Yes, "The Donald" is nbCBoth parents were U.S. citizens at the time of his birth–Mom had "naturalized"
I’ve recently started a website, the information you offer on this website has helped me tremendously. Thank you for all of your time & work. “Cultivation to the mind is as necessary as food to the body.” by Marcus Tullius Cicero.
It’s a pleasure to find someone who can think so clearly
But yes, the big difference between Poles and Germans is that Poles have a lot less affinity to Africa and Asia.See Table 1. and Figure 2. in this current study.It’s something I’ve always suspected, and now its been proven by huge samples from both countries.
Yes, I am a nurse… Found a patient in her home in full cardiac arrest.. Got her heart started, gave her an aspirin, squad arrived. She’s in hospital now. They just put 3 stents in. She will be ok…
Most help articles on the web are inaccurate or incoherent. Not this!
I would love to hear from you and make this site a fun place for Deviled Eggs lovers. Please share your experience and let us know how we can improve.Thank you
The ability to think like that is always a joy to behold
After study a few of the blog posts on your website now, and I truly like your way of blogging. I bookmarked it to my bookmark website list and will be checking back soon. Pls check out my web site as well and let me know what you think.
If not for your writing this topic could be very convoluted and oblique.
If information were soccer, this would be a goooooal!
C'est vrai que les bretons lisent beaucoup ! Mon père et ma grand-mère sont bretons et ils adorent lire ! C'est d'eux, que vient mon addiction !
Superb blog! Do you have any recommendations for aspiring writers? I’m hoping to start my own site soon but I’m a little lost on everything. Would you recommend starting with a free platform like WordPress or go for a paid option? There are so many choices out there that I’m completely overwhelmed .. Any tips? Thank you!
Itse käytän The Vital Imagen puhdistusainetta ja sillä lähtee kyllä mineraalimeikit tosi hyvin. Ne itseasiassa lähtee hyvin melkeinpä pelkällä vedellä. Muistelin vaan kun täällä jokin aika sitten keskusteltiin siitä miten Hauschkan tuotteilla ei tule puhdasta jälkeä. Ajattelin että yksi ongelma voi tosiaan olla tuo talkki, jota on välillä vaikea saada kunnolla pois rajummillakaan aineilla.
48c“à °°à °šà °¯à °¿à °¤ à °Âà °®à ±€ à °®à °¾à °Ÿà ±Âà °²à °¾à °¡à °¡à ±Â. à °‰à °ªà °¨à ±Âà °¯à °¾à °¸à °¾à °²à °¿à °µà ±Âà °µà °¡à ±Â. à °—à °—à ±Âà °—à ±‹à °²à ±Âà °ªà ±†à °Ÿà ±Âà °Ÿà °¡à ±Â. à °ªà ±Âà °°à °¶à ±Âà °¨à °¿à °‚à °šà °¡à ±Â. à °•à °¾à °¨à ±€ à °† à °ªà °¨à °¿ à °®à °¨à °šà ±‡à °¤ à °šà ±‡à °¯à °¿à °¸à ±Âà °¤à °¾à °¡à ±Â!”“à °’à °• à °¹à °¾à °¯à °¿à °¨à °¿, à °µà ±‡à °¦à °¨à °¨à ± à °’à °•à ±‡à °¸à °¾à °°à °¿ à °•à °²à °¿à °—à °¿à °‚à °šà ±‡ à °Âà °¾à °µà °¾à °¨à ±Âà °¨à °¿ à °Âà °°à °¿à °‚à °šà °—à °²à °—à °¾à °²à °¿.” à °¬à °¾à °µà ±Âà °‚à °¦à °‚à °¡à °¿ à °ªà °°à °¿à °šà °¯à °‚.à °¤à °ªà ±Âà °ªà °• à °•à ±Šà °¨à °¿ à °šà °¦à °µà °¾à °²à °¨à ±Âà °¨à °®à °¾à °Ÿ..!à °¨à °¾à °•à ±Âà °•à ±‚à °¡à °¾ à °¬à °§à ±Âà °§à °•à °‚ à °µà °¦à °¿à °²à °¿à °‚à °šà ±Âà °•à ±Âà °¨à °¿ à °®à °°à °¿à °¨à ±Âà °¨à °¿ à °®à °‚à °šà °¿ à °ªà ±Âà °¸à ±Âà °¤à °•à °²à °¨à ± à °—à ±Âà °°à °¿à °‚à °šà °¿ à °°à °¾à °¯à °¾à °²à °¨à ±‡ à °‰à °¤à ±Âà °¸à °¾à °¹à °¾à °¨à ±Âà °¨à °¿ à °•à °²à °¿à °—à °¿à °‚à °šà °¿à °‚à °¦à °¿ à °®à ±€ à °ªà °°à °¿à °šà °¯à °‚..:)
Pode ser uma idiotice o q eu vou perguntar, mas… Vamos lá…Bah posso substituir o arroz pela quinua em alguma refeição.ADORO SEUS POSTS…[]
Short, sweet, to the point, FREE-exactly as information should be!
I thank you humbly for sharing your wisdom JJWY
Vilken hääääääärlig utlottning, jag vill gärna vara med i den:))) Jag skulle gärna vilja vinna lampan krebs pompe 27 Kristallampett cognac, gah så läcker!!! Jag har länge funderat över vilken vägglampa jag ska ha på väggen i vardagsrummet invid trappen och en sådan skulle vara heeeeeeelt klockren:)))Hoppas på tur!!!Kraaaaaaaaaaaaaam aka
That really captures the spirit of it. Thanks for posting.
You’re on top of the game. Thanks for sharing.
Le référencement est un secteur difficile, les enjeux sont importants et la concurrence est très grande. Je suis nouveau dans le domaine, mais il me semble déjà que sans la passion du métier, il sera difficile à tout professionnel de persévérer. Courage donc à tous!
Thank you for posting this blog and allowing us all to remember JP. Her Spirit is remarkable and I can still see her bright smile. I loved being able to send her Sour Gummy Vitamins… ah, the wonderful memories! Thank you.
Hey, that’s powerful. Thanks for the news.
The Pippa’s Butt Workout is awsome.My butt hurts but it feels great!Also at 2:55 in the video in the background you can see 2 double deckers;on the first one is a comercial with Ted.You should watch this movie(comedy).It’s really funny;))
I see, I suppose that would have to be the case.
Howdy there,I find out that your blog is quite educational and useful and we were interested if there is really a possibility of getting More stories like this on your site. If you willing to support us out, we can be willing to compensate you… Kind regards, Wm Gradert
Pleasing you should think of something like that
Estimado Arquitecto, tuve algunas dificultades para obtener el documento. lo he Hojeado con “H” y me parece interesantÃsimo, en el momento que lo ojee sin “H” emitiré algún otro concepto, de antemano le felicito por el documento se que no es nada fácil, en lo referente a soldaduras recuerda que cuentas con un amigo, de igual forma felicito a Eternit por confiar y patrocinar este tipo de trabajos.
Ah ok,je savais pas qu’il ressemblait à un modèle Maje!!Il est magnifique par contre plus moyen de mettre la main dessus dans ma taille depuis octobre :’(
I came, I read this article, I conquered.
Smart thinking – a clever way of looking at it.
That’s cleared my thoughts. Thanks for contributing.
I want to express my gratitude for your kind-heartedness supporting persons that require help with this important subject matter. Your personal commitment to getting the solution along became particularly important and has usually encouraged those like me to achieve their desired goals. Your personal interesting instruction indicates so much a person like me and further more to my fellow workers. Many thanks; from all of us.
Kimberly – You know being the girl with no sense of direction or any head for geography what so ever, I need little maps of all the places you go so I can keep up. I’m so confused… guess I need to do some googlemaping.
This was the wedding of Mike and BeLara which you may remember I did their “CILS” (Couples in Love Session) last year in NJ (where Belara is from) now we have their wedding day down in Atlanta,GA (where
Taking the overview, this post is first class
Most help articles on the web are inaccurate or incoherent. Not this!
On peut allumer des dizaines de bougies à partir d’une seule sans en abréger la vie…On ne diminue pas le Bonheur en le PartageantJoyeuses Fêtes de fin d’année!Mélodie
Great tutorial – I just love your work! One item I re-purposed was an old wine rack I won on ebay. I filled it with clear plastic cups and store my pens, markers, and little scrapbooking toys in the them. Thanks for the chance to win!
Knowledge wants to be free, just like these articles!
I'm beginning to get a bit hesitant around independent films. So many that are good come to a sudden grim ending.I really liked how it flowed though. He gets a supernatural item. The first major use of it is to steal from a vending machine. While that's excusable, he soon uses it for genuine crime, and suffers by his own doing.However … he would die in there. Darn grim endings…
Feel better soon! I am going to email you a clip from the Moz show :)As for my weekend, I am hitting up one of my favorite annual rummage sales (and right up the street from my house no less!), going to my first craniosacral therapy appointment, and then I have a costumed barn dance to attend in the evening. I am looking forward to relaxing on Sunday 🙂
We definitely need more smart people like you around.
That’s an inventive answer to an interesting question
Hey, good to find someone who agrees with me. GMTA.
I just hope whoever writes these keeps writing more!
Now that’s subtle! Great to hear from you.
Detta blir någonting att berätta för era barnbarn så småningom. Måste vara en härlig resa ni är ute på.Stackars de 6-åringar som fick lyssna på toa besöket, de kommer aldrig mer att våga gå på toaletten utomlands.
Lol! Well, all that is probably true…I’d still probably get sucked in if I ever make it to the Aloha state.Love these goat pictures. What cuties – well, they’re cute as long as they’re not head-butting you…
There’s nothing better than a well played acoustic guitar… especially if it’s a Vintage Gibson… Welcome to the obsessive, time consuming, addictive world of blogging. Once you blog, you’ll never go back.Next is the wonder of Vlogging… Even more addictive!
I usually have meatless meals at least once a week so I could make a more conscious decision and go for a Monday. Your fuscilli dish looks delicious Mary.
Yo, that’s what’s up truthfully.
"In contrast, while there's are lots and lots of things I don't know, there isn't all that much about which I like to boast that my ignorance makes me a better person than the unignorant."If I hear one more person bragging about not speaking any foreign languages, I will scream. And it's worst among the self styled intellectuals among the paleocons. Stop bragging about what you don't know – unless I can boast about being indifferent at maths?
That addresses several of my concerns actually.
14.10.2012 at 07:26 · I just want to mention I am beginner to blogging and honestly loved your web-site. Likely I’m want to bookmark your website . You amazingly have exceptional articles. Thanks for revealing your blog.
I much prefer informative articles like this to that high brow literature.
For a major photography magazine, they hardly have any photos on their website. All they offer are a few measly flash galleries. That, my friends, is the epitome of arrogance!
J’ai Peaches & cream depuis la semaine dernière grâce à vous et pour fêter le retour du soleil et des tong, je l’ai posé sur mes petits pieds, j’adore !!! Juste assez flashy pour l’été et le côté crémeux adoucit le tout ! 🙂
That’s a well-thought-out answer to a challenging question
I want to send you an award for most helpful internet writer.
Thank you for writing about the victim. I feel this troubled(killer)actor is getting too much attention, like most criminals do, whereas the victim gets lost in the story. I feel bad and very sorry for her family and friends, she did not deserve to die in this manner after helping so many people in her life time.
I can’t hear anything over the sound of how awesome this article is.
This is a neat summary. Thanks for sharing!
Residenza is the last place someone should look for a home. You work with illiterate, lazy and unorganized people, who make your life hell. We have been suffering for the past 1 year on this project. and we have given up the flat because we were about to run in the flat and hammer up all of their work- it was that bad. DO NOT BUY IN RESIDENZA OZONE. USE YOUR MONEY, TIME, and EFFORT SOMEWHERE USEFUL! we did not know how bad this place is, we don’t want people to suffer like we did.
That really captures the spirit of it. Thanks for posting.
Le mie donne preferite? Tutte quelle che mi piacciono ^_^A parte questa ovvietà , se devo proprio indicare un periodo storico-cinematografico, direi quelle degli anni '70.Ridatemi la Fenech e la Gloria Guida di quegli anni. Mai visto nessuna donna più arrap… SENSUALE.
Wonderful job right here. I truly enjoyed what you had to say. Keep going because you undoubtedly bring a new voice to this topic. Not many people would say what youve said and still make it interesting. Nicely, at least Im interested. Cant wait to see much more of this from you.
You’ve got it in one. Couldn’t have put it better.
je suis POUR une interdiction de la vente à découvert à nu (pour tous les titres, pas seulement les obligations publiques), à ne pas confondre avec une interdiction de la vente à découvert. D’autant plus qu’une interdiction de la vente à découvert à nu est une proposition qui a de fortes chances d’être adoptée par de nombreux pays (quand ce n’est pas déjà le cas), Etats Unis compris et sur laquelle il devrait être facile de se mettre d’accord. C’est d’ailleurs ce que propose l’Allemagne.
Hey AB – I would say "Did you bother to read what I wrote?" but this is you, so the answer is 'no'. I agree that Obama is a corporatist. Do I need to be any clearer? Do I need to finger-paint it on cave wall for you? Until you start thinking clearly, leave me alone. Grown folks are talking.
I am often to running a blog and i actually recognize your content. The article has really peaks my interest. I’m going to bookmark your site and keep checking for new information.
That’s not just logic. That’s really sensible.
A really good answer, full of rationality!
Mormon to tez czlowiek jak katolik.Widac ze czas wymienic swoje „klepki” na cos madzrejszego i nie pieprzyc glupot. Oby tylko nie Obama i ten Biden na nastepne 4 lata.
An interesting discussion is worth comment. I think that you should write more on this topic, it might not be a taboo subject but generally people are not enough to speak on such topics. To the next. Cheers
Way to go on this essay, helped a ton.
Frankly I think that’s absolutely good stuff.
I’m so glad that the internet allows free info like this!
Thank you so much for the updates, Empress. Sending prayers to Diva and fam for a speedy recovery. Thought of Diva tonight while glitter spraying my nephews wasp wings…I know Diva’d be all glittered up get well soon! <3
you are a blogger who attempts to make money online completely through Google AdSense, writing paid reviews, or even by selling advertising blocks, you are probably missing out on a
giovanni scrive:Interessante i vari suggerimenti di questo libro. Cercero’ di farne tesoro, e mettere in pratica la teoria espressa in questa recensione. Un saluto al dott. R: Ciruolo per la sua valida collaborazione. Giovanni A.
Your post has moved the debate forward. Thanks for sharing!
annuler mon rdv ou assumer et voire se q il v on me dire ,je vous avoue que je suis un peut perdu ,je ne fuit pas la sanction ,mes je redoute quil me supprime le permis !!!!!!! je l assume mes bon !!!!
I’ve learn a few good stuff here. Certainly value bookmarking for revisiting. I surprise how much effort you put to create any such great informative web site.
I like getting the letters, but please really don’t go telling folks they are going to receive about a single a week. We have been lucky for getting one a month. You need to do a little better than that….or cease the pinocchio stories at the pretty least. If just one a month is what it is, then that is what it is.
Generally I don’t learn post on blogs, but I wish to say that this write-up very pressured me to try and do so! Your writing taste has been surprised me. Thank you, very nice article.
, March 1, 2011 at 6:20 am Unfortunately Ashley, we will only be offering ad space to local business owners from our area. Links are always an option though:) Let me know if you'd like to reciprocate links.
They had my favourite cereal back in after ages, though (the strawberry and almond one), and I got a pretty good deal on underwear, so…I can’t stay mad at them. I didn’t get such surly service, though.
“à ®…à ®£à ¯Âà ®£à ®¾ à ®¹à ®šà ®¾à ®°à ¯‡ à ®Žà ®©à ¯Âà ®•à ®¿à ®± à ®‡à ®¨à ¯Âà ®¤ à ®‡à ®¨à ¯Âà ®¤à ¯Âà ®¤à ¯Âà ®¤à ¯Âà ®µ à ®•à ¯‹à ®®à ®¾à ®³à ®¿à ®¯à ¯ˆ….” à ®•à ¯‹à ®®à ®¾à ®³à ®¿ à ®…à ®²à ¯Âà ®². à ®…à ®£à ¯Âà ®£à ®¾ à ®¹à ®šà ®¾à ®°à ¯‡ à ®’à ®°à ¯ à ®ªà ®¯à ®™à ¯Âà ®•à ®°à ®µà ®¾à ®¤à ®¿à ®¤à ®¾à ®©à ¯Â. à ®‰à ®³à ®µà ®¿à ®¯à ®²à ¯ à ®¨à ®¿à ®ªà ¯Âà ®£à ®¤à ¯Âà ®¤à ¯Âà ®µà ®®à ¯ à ®µà ®¾à ®¯à ¯Âà ®¨à ¯Âà ®¤à ®µà ®°à ¯Âà ®•à ®³à ¯ à ®…à ®µà ®°à ®¤à ¯ à ®®à ¯Âà ®•à ®¤à ¯Âà ®¤à ¯ˆ à ®ªà ®Ÿà ®¿à ®¤à ¯Âà ®¤à ¯Âà ®ªà ¯ à ®ªà ®¾à ®°à ¯Âà ®™à ¯Âà ®•à ®³à ¯Â. à ®‰à ®£à ¯Âà ®®à ¯ˆ à ®ªà ®³à ®¿à ®šà ¯Âà ®šà ¯†à ®© à ®ªà ¯Âà ®²à ®ªà ¯Âà ®ªà ®Ÿà ¯Âà ®®à ¯Â.
A bit surprised it seems to simple and yet useful.
I was suggested this website by way of my cousin. I am now not positive whether thissubmit is written through him as no one else recognise such certain approximately my trouble.You’re amazing! Thank you!
Um ok WOW!Just a sec, let me rephrase that…WOW WOW! That is so freakin cool! Where did you get your ideas? You are so good at men's cards, I so aspire to be just like you but alas…I am not. So sad for mini-me 2. You just have to win something on this!SSOA x a million!
What a joy to find such clear thinking. Thanks for posting!
If you wrote an article about life we’d all reach enlightenment.
Let’s use Kuliah Gratis to maximize your skill in manage vcctermurah , don’t forget to create and improve something like and great opportunities will be given in Kompetisi Website Kompas MuDA-KFC I also have many view about this articles, see in Kompetisi Website Kompas MuDA-KFC With these in my blogs, I must go to Kompetisi Website Kompas MuDA-KFC When your realizes handphone nokia terbaru , you will find a lot berita kompas terbaru and let's belajar akuntansi keuangan
Una cosa che mi ha fatto ridere è stata la mini sega che spunta tra gli orpelli del braccio bionico. La lama gira a due all'ora. Un Silkepil mi avrebbe fatto più paura. Ad ogni buon conto, da utilizzatore di programmi 3D per lavoro, non posso fare altro che applaudire al progetto. Bravi tutti.Ma ve l'immaginate un epilatore a due cm dalla faccia e la barba un po' incolta? Brrrr…
This piece was cogent, well-written, and pithy.
Kaneganese,Thank you for the translation. That is similar to what I also was thinking; however, in the context of the passage, I think å€èˆ¡æ¼æŽ¢è€…時到 should be translated as «from the time Japanese boats came to fish, they have called the island ‘Takeshima.'» What do you think?
I Google+'d the post too!I also realized I did not leave a valid email address in my comments, so here it is….mommyinstincts AT gmail DOT comhope that counts…if not, let me know and i will go back and delete all my comments and start over! LOL I'm desperate to win this for lily, so i WILL do it! 😉
ai Denise, essa saudade vai e volta, em cÃrculos, uns dias ela está suave, outros é terremoto, vulcao e tsunami juntos. Nos tira deste mundo, fica um mixto de dor, revolta, tristeza no nosso ser, para depois passar e nos fazer olhar o mundo com outros olhos.um abraco
This insight’s just the way to kick life into this debate.
Merci Christian d’avoir pris le temps d’écrire cette longue explication, je pense que cette mise au point était vraiment utile.Merci d’avoir pris le temps de le faire!
Zdravim.V knize Domácà výroba lihovin popisujà recept ze Å¡krobnatých surovin takto:obilà semlÃtohřát trojnásobné množstvà vody na 90C a smÄ›s zalÃt,po dúkladném promÃsenà musà mÃt 70Cupravit PH na 5 a pÅ™idat pivovarský slad nebo amylolytický enzymu teploty 27C pÅ™idat enzym ovopres a kvasinky.kde by se dalo sehnat amylolytický enzym nebo jaká by mnÄ›la být dávka sladu nevÃm, upravu PH by Å¡lo upravit kys. citronovou.Pokud by mÄ›l nÄ›kdo upÅ™esňujÃcà info múžete múj recept doplnit.
Your pictures are really extraordinary and they are a joy to see. This one was already outstanding out of the camera.It is a gift to know how you work. The story is shocking and really interesting and the other pictures are also amazing.Many thanks for sharing and congratulations for your work!
Super excited to see more of this kind of stuff online.
Eu amo filmes de cavalo e esse meu segundo preferido o primeiro e secretariet + esse e muito bom ,bom mesmo e eu amei muito …!!!…e tbm amo cavalos pretos!!!na verdade amo tordos os cavalos de todas as raças e cores
You should include the Horse Drinking Thorough for the Romans because it would change the cavalry’s attack points for Romans dramatically. It would make both the Equites Imperatoris and the Equites Caesaris have the same amout of attack value as the axemen, but there is still crop cunsumption so it would’nt be wise
“And if you don’t plan doing it, why are you preaching to others?”But we’re only preaching to you that you do what we did… give them the vote and be a state of all your people.
Só um imbecÃl mesmo pra vestir uma camiseta da Dilma travestida de Guevarra ou é o Guevarra travestido de Dilma, porque não protestam pela prisão da Blogueira cubana? Eu quero que estes comunas de merda vão tudo para o quinto dos inferno!
Right on-this helped me sort things right out.
Comfortably, the post is during truthfulness a hottest on this subject well known subject matter. I agree with ones conclusions and often will desperately look ahead to your . Saying thanks a lot will not just be sufficient, for ones wonderful ability in your producing. I will immediately grab ones own feed to stay knowledgeable from any sort of update versions. get the Work done and much success with yourbusiness results!
I’ll try to put this to good use immediately.
In awe of that answer! Really cool!
qui aime les « mots simples » voici pour vous:« Toi qui cherches la véritéc’est le mur que tu cherches.La pierre l’autre pierre l’autre.Appuie ton dos sur le murou ton frontreste ainsi debout dans la nuitavec les petites pièces de monnaie dans ta pochequi ne rendent aucun sond’or ou d’argentsans aucune valeur.les rêves non plus. » Yannis Ritsos, Athènes 1973, traduction de Dominique Grandmont
Your article perfectly shows what I needed to know, thanks!
I believe that a property foreclosure can have a important effect on the applicant’s life. Mortgage foreclosures can have a 6 to ten years negative impact on a debtor’s credit report. A borrower who’s applied for a mortgage or any kind of loans for that matter, knows that a worse credit rating is definitely, the more complicated it is to obtain a decent loan. In addition, it might affect the borrower’s power to find a decent place to lease or hire, if that becomes the alternative houses solution. Good blog post.
That saves me. Thanks for being so sensible!
Fábio Ricardo • 28 de Fevereiro de 2012 às 03:24Vi uma palestra do Fábio Ricotta em que ele teceu diversos elogios a este plugin.Instalei, mas fiquei meio perdido sobre como fazer a instalação correta.Esse post é perfeito, tirou todas as minhas dúvidas e resolveu no passo-a-passo.Parabéns e muitÃssimo obrigado!
The honesty of your posting shines through
Smith now has something besides VD and Crabs to get his balls to. He needs to stay Smith is at least a known commodity. you have receivers for him. Sign him and respect the season he gave you last year. Manning is one hit away from being paralyzed (I guess they all are, but he is more of a china doll than the rest).
I’ll immediately grasp your rss feed as I can’t in finding your email subscription hyperlink or newsletter service. Do you have any? Kindly let me understand so that I could subscribe. Thanks.
Thanks for starting the ball rolling with this insight.
Il ne faut pas se leurrer, ce n’est pas une victoire face aux verts qui sauvera Puel. Contre Sainté, encore plus à Gerland, même le match nul est interdit. Crise ou pas crise.Ce que peut faire Puel à l’OL ne se jugera pas sur 5 ou six matchs, mais sur ses 2 années passées au club.
That’s a cunning answer to a challenging question
it already but …This blog rocks! I gotta say, that I read a lot of blogs on a daily basis and for the most part, people lack substance but, I just wanted to make a quick comment to say I’m glad I found your blog. Thanks, A definite great read..Tony Brown
How do you know it was the motherboard that fried? It could be any part of the computer like RAM, the chipset or even the CPU!Most of the time its either the RAM or the chipset that went bad!!!Love the channel and the great videos you post.Keep up the amazing job with more of your videos =p
Solano: Haga los ejercios que quiera…y yo los comentarios que quiera. Asà es la democracia.Al otro: Una violación no justifica la otra. El territorio de Ecuador fue violado varias veces por Colombia. Es raro hacer justicia cometiendo injusticias.
, I also pray that he will make the most of his opportunity in the ‘Elephant Room’ to make sure that T D Jakes not only clearly defines his beliefs about the Trinity, but that in brotherly love and by the power of the Holy Spirit, he will allow as much time necessary to share Scriptures that will make the doctrine of the Trinity as clear as possible. God bless you and your ministry.
Nou dat klinkt goed! Wij haalde in Italië ook vaak onze broodjes en beleg in de supermarkt, om het onderweg ergens op te peuzelen. Scheelt een hoop geld iid! En moet zeggen dat ik het ook wel leuk vind om de lokale supermarktjes van binnen te bekijken .
Yes! I have been very impressed with what I’ve seen with the S4 so far. I can’t wait to get rid of this BB Bold 9700 but I’m still waiting to see what Sammy has for us on Thursday before I make a decision on my first Android phone. I live in Toronto and we’ve had the One X available for a few weeks now. It’s been tempting but I decided to hold off to see how the tegra3 and S4 compare. Now its just a matter of what Samsung does on Thursday and if they will have the Galaxy S3 released here in a reasonable time frame. I’m getting tired of waiting. The Snapgragon S4 will do just fine. No Tegra3, no problem.
jeg husker også hvordan vi fikk inn skychannel. vi fikk tidlig parabol og den var ganske stor der den stod, den var såpass at solen forsvant ved fire tia på tomta vår om sommern.. vi solgte den, du kan forresten se den på venstre side når du kjører motorveien i sverige om du feks skal til gøteborg igjen snart.. line.
Hiya, I’m really glad I have found this information. These days bloggers publish only regarding gossips and internet and this is really frustrating. A clear locate with attractive subject matter, this is what I need. Recognition for keeping this website, I’ll live visiting it. Perform you achieve newsletters? Can not get hold of it.
I’m so sorry about the catpcha, Hazel. I tried to disable/remove it, but it came right back! I need a little techy help, need to enlist son’s (or someone wordpress.org familiar). If it’s any consolation, it’s also a big trial for me, as my vision is worsening with Fuchs’ dystrophy. We’ll get it fixed, eventually!Meanwhile, thank you so much for making the difficult effort to leave this good comment.(Now here goes my repeated recaptcha effort…)
exact: “PregăteÅŸte cineva fermieri în Å£ara asta”???ii inveti sa se asocieze si sa-si faca ferme functionale? sau ii presezi sa renunte la terenuri pe doi bani?sau: tie, tovaras sau domn stat-privat, cat iti iese in fiecare varianta?
old bogus, Yes Lisa is correct in her comment. It seems that the support for Obama is as mindless as the supporters of the kkk. That’s all i’m trying to say. In my world that is a fact.As for Obamas legitimacy , you can judge for yourself. This is none of my concern. jim
Thank you sweetheart. You are a great big bright light in my life! So grateful for our soul connection sweet sister. Love you!! p.s. still waiting for you to share your wisdom, love & light in those angel cards
I simply want to tell you that I’m all new to blogs and truly enjoyed this page. Most likely I’m likely to bookmark your site . You amazingly come with really good stories. Thanks a lot for sharing with us your webpage.
Howdy very nice website!! Man .. Excellent .. Wonderful .. I’ll bookmark your blog and take the feeds additionally…I am happy to search out so many helpful info here within the publish, we want work out more strategies on this regard, thank you for sharing.
That’s cleared my thoughts. Thanks for contributing.
Consulenti WordPress | Shophouse Platinum I was suggested this blog by my cousin. I’m not sure whether this post is written by him as nobody else know such detailed about my difficulty. You’re wonderful! Thanks! your article about Consulenti WordPress | Shophouse PlatinumBest Regards Shane
Hej Birgitte,Hvor er det flot med dit vægttab! Nu kender jeg ikke Dukan-kuren særlig godt, men jeg går ud fra, at der nok er andre forudsætninger i den kur, der gør, at det ikke er nødvendigt for dig at spise bønner eller linser. Bønner og linser er kulhydrater og kulhydrater hjælper din krop med at udnytte din kost ordentligt mht fedt og proteiner.Hilsen Sanne
doctorCondivido il tuo intervento.In special modo ove scrivi: “Se si vuole capire e intervenire occorre provare a pensare gli uomini in modo più complesso.“.Mi pare di intuire, dal nick e dall’ultima osservazione, una tua conoscenza diretta, professionale, delle (come le chiami tu) nuove conoscenze prodotte dalle scienze.Che ne dici di un tuo contributo più esteso? Sempre se sei d’accordo.
Voilà ! c'est fait ! au lieu du poulet grillé barbecue (ah la la , pourvu que ce soit encore l'été dimanche prochain !) , j'ai donc fait les gaufres avec le sucre perlé de Gand et la vieille gaufrière qui va sur le gaz : mortel ! et tout aussi délicieux froid , ce matin , à la pause avec un petit chocolat … Merci !
Szanowny Kolego (takÄ… mam przynajmniej nadziejÄ™)”wkÅ‚ad-nie-nakÅ‚ad”!Ta witryna jest stworzona wÅ‚aÅ›nie po to, aby wszyscy, TY również, mogli wyrazić na niej swojÄ… opiniÄ™. Prosimy tylko, aby zamiast „ble,ble,ble” i osobistych „wycieczek”, byÅ‚y KONKRETY. W MIARĘ KRÓTKO, ZROZUMIALE i „dla ludzi”, a nie rozprawa doktorska.
Hey hey hey, take a gander at what’ you’ve done
That’s a well-thought-out answer to a challenging question
Lauren – Erica, I just love the photo you took of Tiffany and Rob in that field of lavender (July 13, 2011). That’s exactly the place and the kind of picture I’ve been looking for! We’re getting married next summer, hopefully in a location like that. Would you mind sharing where that photo was taken? Was it on Lavender Farms’ property? Also, how much do you charge for a day?Thanks so much,Lauren
I told my grandmother how you helped. She said, «bake them a cake!»
If only there were more clever people like you!
amoreee! que bom ler noticias suas! adorei! continua postando pleaseee! fotos, textos, videos, tudo que puder pra diminuir um pouco a saudade e ver que você está bem e feliz por aÃ!!! aproveita!beijaoooooo
Is there any way to install MySQL in the iPod Touch or iPhone? I been searching some kind of clue but i found nothing. Perhaps someone knows how or can think how to install it. Thanks in advance.
That’s a mold-breaker. Great thinking!
Jag tycker du är supercool, bara riktiga vinnare har tillräckligt med självdistans och självförtroende till att "avslöja" sina fotoknep!Gillar dig OCH din blogg! Ha en bra helg!
Bonsoir ravissante demoiselle,je me présente Cédric 25ans de Toulon, tres coquin, attentionné,…si tu veux profiter de la vie sans te prendre la tête vien me voir :je peu recevoir, je fait aussi les massages,…Bisoux et peut être a bientôt
I like to party, not look articles up online. You made it happen.
Thank you so much for this article, it saved me time!
Perfect application and great tips I am usually too lazy and skip the cleanup step, but my hand has become more steadier so I don’t usually get the polish on my cuticles anymore.
The genius store called, they’re running out of you.
oi Zel! Eu sou aquela que ficou com vergonha de te cumprimentar no Etna… hahaahha!Menina, e o que é a ditadura da chapinha???? Porque as mulheres nao assumem os cachos? eu tenho cabelo cacheado e adoro, aprendi a lidar com eles (nao é qquer cabelereiro que sabe cortar e cuidar) e não aliso meus cachinhos por nada nesse mundo.beijoca!
If you’re looking to buy these articles make it way easier.
How could any of this be better stated? It couldn’t.
Wow! Did I say WOW?!? There was never any doubt that Lexi is an exquisite beauty, but your ability to bring out the best in her is well evidenced by all of these outstanding senior portraits. She is a very lucky girl indeed. Beautiful girl, beautiful work…just beautiful.
Wham bam thank you, ma’am, my questions are answered!
Lorenna Karolynne disse:Haidêe adorei seu blog, está de parabéns, assim como em tudo q faz, sempre c seu capricho e carinho. Adoro teu trabalho, vc nasceu c o dom. Tudo mt perfeito!Bjos! Lorenna
than short produces less emphasis than those…that vary sentence length. longer sentences should explain ideas, while short sentences should intensify those ideas.• begin or end with key points – the main topic of a sentence should never get consumed by the context. ideally, you should place …
Superb read, I just passed this onto a friend who was doing a little investigation on that. And he actually bought me lunch because I located it for him smile So let me rephrase that: Thanks for lunch!
Je n’ai aps le son au boulot( pfff) je vais �couter la vid�o tout � l’heure ! D’une mani�re g�n�rale je trouve que Julien Dor� est un peu + artificiel que Willem, nu peu plus «travaill� dans le style.
Woot, I will certainly put this to good use!
puff pastry would be fine. you could probably pour cake batter over top and bake like a crumble. it’s all in the wonderful caramel that becomes of the sugar and apple juices.i baked at 350 and at 425 and they both worked fine–just cook it till it’s nicely browned. it’s the bottom of the tart.i used between 6 and 8 apples. the more apples you get into the pan the better.
Sicherlich kannst Du im Gasbackofen Chips trocknen. Ich denke sie werden dann vielleicht nicht mehr Rohkostqualität (bis 42 Grad) haben, aber sie sind allemal vieeeeel gesünder als alle anderen Chips die es in Tüten zu kaufen gibt Ich liebe es auch, die Chips als Crunchies über den Salat oder die Suppe zu geben…liebe Grüsse von Conny
c1f1Hey I am so excited I found your site, I really found you by error, while I was browsing on Google for something else, Regardless I am here now and would just like to say thanks for a marvelous post and a all round interesting blog (I also love the theme/design), I don’t have time to go through it all at the moment but I have book-marked it and also added your RSS feeds, so when I have time I will be back to read much more, Please do keep up the superb work.ca
Bilderber 2011: EU Fall Threatens One World Currency | The Line in the Sand Excellent goods from you, man. I’ve understand your stuff previous to and you’re just extremely wonderful. I really like what you’ve acquired here, really like what you’re saying and the way in which you say it. You make it enjoyable and you still take care of to keep it wise. I can’t wait to read much more Bilderber 2011: EU Fall Threatens One World Currency | The Line in the Sand again from you. Thanks For Share .
..non è il COBOL a gestire gli anni con solo due cifre nelle date, ma è stata una scelta della maggior parte dei programmatori per risparmiare memoria. bisogna tener conto che fino a qualche anno fa i pc non avevano grandi risorse di ram.
Quando un leghista che ha un partito che salva degli indagati per associzione mafiosa (non solo Romano; ma ad esempio Cosentino) etc etc dice che Fini è il peggio del peggio, vuol dire che si è sulla strada giusta
One thing you could also include in your diet is to drink lots of water — so you won’t be drinking as much snapple or soda, even if they are diet. I think the healthiest really is water.
Such a cool and different campaign, really love the pictures!I mainly read classic novels during my high-school time and my favorite is "Great Expactations" from Charles Dickens!Have a lovely Easter weekend!
YMMD with that answer! TX
To think, I was confused a minute ago.
diyor ki:Site başka siteden ilanları çekiyor fakat direk anlaşmalı emlak firmaları da var. Sadece koordinatlı ilanların bulunması için bence çok güzel bir site ama yasallık ve içeriğin güçlenmesi zaman alacak gibi. Ama ben kesinlikle ziyaret ediyorum.
Deep thinking – adds a new dimension to it all.
Haha. I woke up down today. You’ve cheered me up!
I knew I wouldn't fit in with the clay breaking crowd when I got the stinkeye from some guy because his Caesar Guerini double almost got hit by a shell tossed from my 1100. Hey buddy, it's an auto, it does the work for me. If your gun's finish is too delicate to be touched by a flying empty shell, either stand to my left, leave it in the safe, or stop blaming me you paid too much for the wrong gun.And stop putting your muzzle on your foot! Are you really willing to destroy your toes?
That’s an astute answer to a tricky question
The beauty of the women in our family still astounds me!!! Thank you for sharing these amazing photos, I am posting them on mt fb page! Gorgeous couple! nuch love and congratulations to you both. Teresa Foster
I like a meal at home that I've cooked for my hubby. Some nice chocolates are absolutley essential too!I wouldn't thank anyone for fresh oysters either, but chocolate ones with pearls are very clever.
Smart thinking – a clever way of looking at it.
I feel satisfied after reading that one.
Poor girl – I hope you get some answers soon. I know from experience that living that way is no fun at all.Please advocate for yourself and get some answers.
Thanks for sharing. Your post is a useful contribution.
You could certainly see your enthusiasm within the work you write. The world hopes for even more passionate writers like you who are not afraid to mention how they believe. Always follow your heart. “Every man serves a useful purpose A miser, for example, makes a wonderful ancestor.” by Laurence J. Peter.
And I thought I was the sensible one. Thanks for setting me straight.
Whoever wrote this, you know how to make a good article.
I guess finding useful, reliable information on the internet isn’t hopeless after all.
Wheat really has little nutritional value. It can have fiber if eating whole wheat, but you get more fiber, pound for pound, from any part of the vegetable that grows above ground. The only reason it has signicant vitamins is because it is fortified by millers who add vitamins to it. We’ve recently learned that athletes, arguably the healthiest of us, remove gluten from their diets when training to reduce the inevitable inflamation caused by severe training. The tells me gluten is bad for everyone. It’s a matter of degree, but how much inflamation supporting food should anyone eat?
Hi, Really cool snack:-) I don’t like pineapple, but any other fruit juice too would be a great combo with masala nuts. Very nice entry!
Me encanta combinar lo sencillo con lo exuberante, el blanco con el oro, es algo asà como un minimalismo barroco?Aquà seguiremos este mesecito de calores, disfrutando de tus propuestas.
Yup, that’ll do it. You have my appreciation.
Tara, i’m surprised you don’t have alot of people commenting on here or on your twitter. I know you have alot fans out there. If i were in Mass. i would have caught your performance, i’m sure it was very good. I’m in the midwest, come to Chicago or St. Louis sometime.
Fotone, ti co vÄ›dÃ, trénujà hlavnÄ› morál. Poslednà dobou (bohužel nebo bohudÃk – teÄ nevÃm, musÃm jeÅ¡tÄ› o tom chvÃli pÅ™emýšlet) mi jde tento trénink skvÄ›lÄ›. HlavnÄ› po obÄ›dÄ›… Ach jo… to je život, jak Å™Ãká můj kamarád. TomasKa
lekker dør Moa ;)sommerfugler er dekorative skapninger og nydelige Ã¥ se pÃ¥ ute i det fri..som jeg gleder meg til sommeren..sitte stille ute og se dem fly forbi ;)ha ei fin uke..deilig med nye blanke ark…klem til deg..Anne♥♥♥
Pretty portion of content. I simply stumbled upon your web site and in accession capital to claim that I acquire in fact loved account your weblog posts. Any way I will be subscribing on your feeds and even I success you get right of entry to persistently fast.
à°…à°¯ిà°¤ే, ఇప్à°ªుà°¡ో à°šిà°•్à°•ు à°•ూà°¡ా వచ్à°šింà°¦ి. à°²ాà°²్à°²ూ à°—ాà°°ు à°¸ైà°¡్ à°¬ెà°°్à°¤ుà°²ు à°…ంà°Ÿే à°ª్à°°à°¸్à°¤ుà°¤ం ఉన్à°¨ à°°ెంà°¡ు à°¬ెà°°్à°¤్ à°² à°¸్à°¥ాà°¨ంà°²ో à°®ూà°¡ు à°¬ెà°°్à°¤ుà°²ు à°‰ంà°¡ేà°²ా à°•ొà°¤్à°¤ à°•ోà°š్ à°²ు ఆర్à°¡à°°్ ఇచ్à°šాà°°ంà°Ÿ. à°‡ంà°• à°ª్à°°à°¯ాà°£ాà°²్à°²ో à°¸ైà°¡్ à°²ోయర్ à°²ో à°•ూà°°్à°šోà°Ÿాà°¨ిà°•ి ఉన్à°¨ à°¸ౌà°•à°°్à°¯ం à°•ోà°²్à°ªోà°¬ోà°¤ుà°¨్à°¨ాం !
It’s imperative that more people make this exact point.
MASYARAKAT BETAWI PASTI MILIH ORANG BETAWI, KALAU SAJA ORANG BETAWI TSB MEMANG PANTAS UNTUK DIPILIH, TAPI KALAU TIDAK PANTAS?? COBA PIKIR LAGI NYET !!!badwordfilter(«4981161972485333735»);
What a pleasure to find someone who identifies the issues so clearly
lum tengok lagi nie…hurrrmmmm jap nak consider…sanggup nak folo ke tak…sbb kalo dah folo…phm phm ajer lah kannnn…
With the bases loaded you struck us out with that answer!
Hi Nickie,So, your board is one of those who are “encouraging” photos. Interesting.I don’t understand why the sellers don’t raise a fuss, too. If I were selling, I’d insist on a fair (maybe even exceptional) chance for my property to be seen.
“à ®•à ¯Šà ®²à ¯Âà ®²à ®ªà ¯Âà ®ªà ®Ÿà ¯Âà ®Ÿ à ®®à ¯€à ®©à ®µà ®°à ¯Âà ®•à ®³à ¯Âà ®•à ¯Âà ®•à ¯Âà ®®à ¯Â, à ®•à ¯Šà ®²à ¯ˆ à ®šà ¯†à ®¯à ¯Âà ®¯à ¯Âà ®®à ¯ à ®‡à ®²à ®™à ¯Âà ®•à ¯ˆ à ®•à ®Ÿà ®±à ¯Âà ®ªà ®Ÿà ¯ˆà ®•à ¯Âà ®•à ¯Âà ®®à ®¾à ®© à ®ªà ®¿à ®°à ®šà ¯Âà ®šà ®©à ¯ˆà ®¯à ¯ˆ à ®‡à ®¨à ¯Âà ®¤à ®¿à ®¯, à ®ˆà ®´ à ®®à ¯€à ®©à ®µà ®°à ¯Âà ®•à ®³à ¯Âà ®•à ¯Âà ®•à ®¿à ®Ÿà ¯ˆà ®¯à ®¿à ®²à ®¾à ®© à ®ªà ®¿à ®°à ®šà ¯Âà ®šà ®©à ¯ˆà ®¯à ®¾à ®• à ®¹à ®¿à ®¨à ¯Âà ®¤à ¯ à ®°à ®¾à ®®à ¯Â, à ®ªà ¯‡à ®°à ®¾à ®šà ®¿à ®°à ®¿à ®¯à ®°à ¯ à ®šà ¯‚à ®°à ®¿à ®¯à ®¨à ®¾à ®°à ®¾à ®¯à ®£à ®©à ¯ à ®ªà ¯‹à ®©à ¯Âà ®±à ®µà ®°à ¯Âà ®•à ®³à ¯ à ®ªà ®¿à ®°à ®šà ¯Âà ®šà ®¾à ®°à ®®à ¯ à ®šà ¯†à ®¯à ¯Âà ®•à ®¿à ®©à ¯Âà ®±à ®©à ®°à ¯Â.”à ®ªà ®¿à ®³à ®µà ¯Âà ®ªà ®Ÿà ¯Âà ®¤à ¯Âà ®¤à ®²à ¯ˆ à ®…à ®´à ®•à ®¾à ®•à ®šà ¯ à ®šà ¯†à ®¯à ¯Âà ®•à ®¿à ®±à ®¾à ®°à ¯Âà ®•à ®³à ¯Â. à ®…à ®°à ®šà ¯Âà ®•à ®³à ®¿à ®©à ¯ à ®šà ¯‚à ®´à ¯Âà ®šà ¯Âà ®šà ®¿à ®¯à ¯ˆ à ®šà ®¾à ®¤à ®¾à ®°à ®£ à ®®à ®•à ¯Âà ®•à ®³à ¯ à ®ªà ¯Âà ®°à ®¿à ®¨à ¯Âà ®¤à ¯Âà ®•à ¯Šà ®³à ¯Âà ®³ à ®µà ¯‡à ®£à ¯Âà ®Ÿà ¯Âà ®®à ¯Â. à ®®à ¯Âà ®¤à ®²à ®¾à ®³à ®¿à ®¤à ¯Âà ®¤à ¯Âà ®µ, à ®ªà ®¿à ®°à ®¾à ®¨à ¯Âà ®¤à ®¿à ®¯ à ®µà ®²à ¯Âà ®²à ®¾à ®¤à ®¿à ®•à ¯Âà ®• à ®¨à ®²à ®©à ¯Âà ®•à ®³à ¯Âà ®•à ¯Âà ®•à ®¾à ®• à ®Žà ®µà ¯Âà ®µà ®³à ®µà ¯ à ®®à ®²à ®¿à ®©à ®®à ®¾à ®•à ®µà ¯Âà ®®à ¯ à ®‡à ®±à ®™à ¯Âà ®•à ®¿à ®µà ®¿à ®Ÿ à ®®à ¯Âà ®Ÿà ®¿à ®•à ®¿à ®±à ®¤à ¯ à ®‡à ®µà ®°à ¯Âà ®•à ®³à ®¾à ®²à ¯Â.
Ana, não percebi bem o teu comentário mas olha que as crianças fartam-se de tomar decisões. Por exemplo, não calçar os sapatos, não comer a papa, não dormir quando dá jeito aos adultos… F, temos, pois, para a troca
I LOVE EVERY SINGLE THING!!!! You look amazing – SO BEAUTIFUL – in your fab nightie, amor! I hope you have the most wonderful weekend – have a cider for me! Love you more than Patti Smith! Sarah xxx
Information is power and now I’m a !@#$ing dictator.
I have been so bewildered in the past but now it all makes sense!
The voice of rationality! Good to hear from you.
thank you SO much for telling me about your experiences. It fascinates me and confirms so much. And yes, I lOVE Hallstatt. I camped there for really cheap, and just a magical place. Hope to go back.
Hi Jayme!Thank you so much for these AMAZING engagement pics and for being there to capture my wedding day. You are such a loving, wonderful person and I am so blessed to have found you! I can’t wait to see the photos from the wedding day! I know they will be magical!-Devon
nice guide, I have read the whole of it and it helped me a lot…can u plz write an guide for travian 4? Also a 3x speed server guide would be very appreciated..thanks
Why does this have to be the ONLY reliable source? Oh well, gj!
Muchas gracias! Realmente soy mucho mas tradicional que lo que expuse en el comentario.. Quizá es que este “café” que tomo por las mañanas y al que llaman café me despierta la ironÃa y el sentido del humor… De nuevo,gracias por tu recomendación Vota el comentario: 0 0
I am totally wowed and prepared to take the next step now.
This is what we need – an insight to make everyone think
These last couple of posts have left me speechless. Are all the local municpal agencies run by ignorant “hillbillies”? (seriously, what is going on? is it in the water?). Thank you for writing these posts and sharing the links. Jane always had a way with bullies. Now they run her security. Still bullies. Just Jane stood them down. (think maffia… the Godmother? running a dynasty, corrupting local officials, sworn to secrecy? “We love all people” is that the front?) Sorry for ranting. Arthur
I’m really into it, thanks for this great stuff!
¿Por qué esa manÃa de querer quitar la clase preferentes?El que quiera ir en preferente que pague un billete más caro (ya lo es) por tener restauración y atención. Pero que no se quite.Con ideas como hacerlo todo turista y eliminar absolutamente todos los servicios hemos llegado a Ryanair. Que si, es todo lo barato viajar con ellos bajo sus condiciones; pero la mafia también dio muchos puestos de trabajo…
Ich mochte früher auch kein Lamm, habe dann allerdings immer wieder probiert, verschiedene Zubereitungsarten versucht. Es ist zwar etwas kräftiger, aber gutes Lammfleisch ist nicht streng. Die Marinde kannst du aber auch für andere Fleischsorten nehmen, wenn es kein absolut kein Lamm sein soll. Huhn oder Kalb passen sicher auch.Gib mal Feedback, ob es geschmeckt hat!
Wonderful blog! I found it while browsing on Yahoo News.Do you have any suggestions on how to get listed in Yahoo News?I’ve been trying for a while but I never seem to get there! Thank you
I totally agree with bak,I like the facade composition too,whereas I would have loved to see balconies too and that s probably why I wouldn t live in there too.
Furrealz? That’s marvelously good to know.
Vicki · Mixed grill with saffron rice is my asparagus salad story. It has been at least two summers since I made this dish that was so amazing that I actually took pics of it (I am not a food blogger so it was a big deal to photograph it). It is so amazing that I feel I need to share it with others the next time I make it.
Oh, I just love this idea! Imagine how much we could keep out of the landfill if we all did this more. I mean, like the entire country. (Sigh.)Well, at least WE’RE doing it, right?
MaCherrie wrote.. SPEECHLESS, amazing dress and the glasses you guys were wearing I love it. Still want to know more about Leigh's glasses.ADR kills the shot.ALFIEhyyp://macherie-macherie.blogspot.com
Looks like a FUN night! And omigosh, that picture of you and your friend behind the Jenga? Hilarious!! 😀 Can I share a secret? I've been planning a big ol' SURPRISE 30th birthday for my husband and it's TOMORROW!! Eeeek. I'm SO nervous (and this is the first time I've shared in blogworld… Doubt he'll find out from my comment haha ;-)) I'm SO going to get funny party drinking things like the goggles, love it!
Yo, good lookin out! Gonna make it work now.
Hi there! This blog post could not be written much better! Looking through this post reminds me of my previous roommate! He always kept talking about this. I am going to forward this information to him. Pretty sure he’s going to have a very good read. Many thanks for sharing!
Thank you so much for this article, it saved me time!
Thanks for sharing. Your post is a useful contribution.
Boy that really helps me the heck out.
Unbelievable how well-written and informative this was.
Wow, that’s a really clever way of thinking about it!
What i don’t understood is in truth how you’re no longer really much more well-liked than you might be right now. You are very intelligent. You realize therefore considerably in relation to this subject, produced me in my opinion consider it from a lot of various angles. Its like women and men aren’t fascinated except it’s one thing to do with Lady gaga! Your own stuffs nice. At all times deal with it up!
Back up to the background of Lafleur-Petrus.. I do believe that before Le Gay and Lafleur were separate entities, they were in fact part of a larger property, one and the same..Le Manoir de Gay. Unless this is complete bollocks that I’ve read!
Also I believe that mesothelioma cancer is a exceptional form of cancer malignancy that is usually found in those people previously exposed to asbestos. Cancerous tissue form inside the mesothelium, which is a safety lining that covers the vast majority of body’s internal organs. These cells typically form inside lining from the lungs, stomach, or the sac which encircles one’s heart. Thanks for giving your ideas.
This information is off the hizool!
eu tenho medo do meu parceiro me criticar e falar mal do meu beijo e me difamar para os outro meninos e ficar com fama de que beijan mau tenho muito medo……………
A rolling stone is worth two in the bush, thanks to this article.
From the NASA proposal they say you could walk across the receiving dish with no I’ll effect. You can’t do that with any of the large telecommunication dishes. The beam will be tightly focused on the receiver and it will probably be a restricted area.
yaya… i hat it many times liow is in klang one..yaya it’s very delicious, i love to eat..yeah..i just only know to eat din managed to take pic..keke then how much do they charge by head?
Nice read, I just passed this onto a colleague who was doing some research on that. And he actually bought me lunch since I found it for him smile So let me rephrase that: Thank you for lunch!
Together with the whole thing which appears to be building inside this particular area, a significant percentage of viewpoints tend to be very radical. However, I appologize, but I do not subscribe to your whole plan, all be it refreshing none the less. It appears to everyone that your remarks are actually not totally validated and in actuality you are yourself not really fully convinced of your point. In any case I did appreciate reading through it.
How neat! Is it really this simple? You make it look easy.
¡Mosquis!, ¿y las chicas no tenemos nada para atinar? Bueno, bueno, que no quiero yo hablar de machismo ahora, esperaré a los retortijones para echarme unos sudokus…Hay que ver…Salu2!!
Hey, I love youtube and all, but i found no other way to contact you than this.The ONLY thing I always complained about youtube is the player, why can't we seek videos' position with little jumps (seconds). I mean, if I want to view second 0:32, maybe the seeking is divided into two parts and i have to view from second 0:13.Thanks for everything, guys!
vraiment sympa en effet ce script 🙂 Mais depuis quelques jours, je suis un peu trop purifié .. tout ma page d’accueil facebook est masqué, je me demandais si ça venait juste de chez moi, juste sur opéra (flemme d’installer un autre navigateur) ou si fesse bouc avait contrer la chose
I’m so glad that the internet allows free info like this!
Looks like you are going to be having some smashing good times ahead! I hate shopping too, I either buy or browse for everything on line so I have to spend as little time shopping for it as possible! I love your pretty card such a cute rainbow scene hugs Rebekah xx
A little rationality lifts the quality of the debate here. Thanks for contributing!
I never thought I would find such an everyday topic so enthralling!
Ciao Riccardo,Con molto piacere vias emailo se hai skype. Il mio contatto skype è mousesenzapalle2007. ovviamente se ti va di aggfiungermi. Se mi dai la tua email, ti mando un’email, se preferisci.
Fantastisk! Jeg gleder meg til å pynte pepperkakehus med min 2 åring senere denne uka! Det blir spennende å se hvor mye som kommer på huset, og hvor mye som kommer i magen!God Adventstid!
You write so honestly about this. Thanks for sharing!
agree with several above that savard at 33 and with 5 or 6 yrs left is a big no for me. rozy has only 2 yrs left and after this yr the real cash of the last yr is 3 mm so he could be easily traded to a team near the cap floor.this team does not need a 33 small center with history of concussions – we already have 1. no need to trade for another with many yrs left on contract
Correction : « dont j’enlève une augmentation constante » doit être remplacé par « dont j’enlève la droite qui ajuste le mieux ». La droite peut descendre, monter, être une constante. Je renvoie au code.Désolé du typo.
You saved me a lot of hassle just now.
Holy shiznit, this is so cool thank you.
Heya i am for the first time here. I found this board and I find It truly useful & it helped me out much. I hope to give something back and help others like you helped me.
ÙÂعلا که از مصاØÂبه خبری نیست اما اگر قبلا تاریخ مصاØÂبه را با نامه دریاÙÂت کرده اید (طبق رویه گذشته) نوبت بعدی نیز نامه دریاÙÂت خواهید کرد البته اگر ÙÂرض کنیم که این رویه تغییری نکند. پس باید آدرس جدید را به دÙÂتر اداره مهاجرت در مونترال اطلاع دهید.
Perfetto Andre, comunque dovrebbero aprire anche una sede dell'E.N.P.A. (Ente Nazionale Protezione Animali) nel medio campidano e sono stato contattato proprio in questi giorni, appena mi è possibile mi avvicino a Cagliari e come ho informazioni ti farò sapere. 0 Mi piace
At last, someone comes up with the «right» answer!
I’ve been surfing online more than three hours today, yet I never found any interesting article like yours. It’s pretty worth enough for me. In my view, if all website owners and bloggers made good content as you did, the internet will be much more useful than ever before.
You’ve really helped me understand the issues. Thanks.
[quote name=»Ã˜Â£Ã˜Â¨Ã™ÂˆÃ™ÂˆÃ™Âˆ صالػ]مشكور أخوي ومجهود جبار وَ يÙÂشكر علية بس ياليت لو كان ÙÂية شرؠللمعابد لأن صراØÂØ© ÙÂية معبد ماقدرت أدخلةالي مكانة مرتÙÂع, وÙÂوقع ثلاثة أعمدة وبجانب المعبد كنز أيضاً لا يمكنك الوصول إليةÙÂـياليت Ø£ØÂد يشوÙÂلي ØÂل :cry:[/quoteشكرا لك اخى على التشجيعبس ياريت توضؠÙÂى أى مرØÂلة هذا المعبد
dogdo-takeshima,Besides, there isn’t any «Dokdo» that you irrationally and blindly fight for as its out-of-pocket propagandist in the BGN database.There is your nickname «Dogdo», however.
Eric, je ne nie en rien tout ça, j'attend juste qu'on me le prouve… Et parfois, j'avoue c'est tellement beau que ça donne envie d'y croire, mais entre l'envie et la réalité il y a un monde 😀
En gång bjöd jag hem vänner för att berätta om Krilon-trilogin av Eyvind Johnson. Något jag tänkt på medan jag läste dem var hur smakligt han beskrev att ta sig en snaps tillsammans med bröd, smör och ost, så det var just det som jag bjöd på.
What a pleasure to find someone who thinks through the issues
Quelques heures de repos suffiront-elles ? Le héros, qui tombe de son fauteuil, a besoin de solides douceurs comtoises, pour répondre à l’appel quotidien des internautes, des « accronautes » devrais-je dire… Merci Martin, pour ces clins d’oeil, qui préparent l’arc – en – ciel, que nous attendons. Nous avons besoin que tu déchires le voile de la sombre actualité, qui pèse, depuis des jours et des jours…
This posting knocked my socks off
Pobre Zita, que outra coisa pode dar um cancro moral se não metástases gosmentas? Ainda por cima com aquele ar de bruxa velha e feia. Tenham pena: para castigo, já lhe basta o espelho.Luis Nogueira
That’s a slick answer to a challenging question
pekala,hepiniz yanılıyorsunuz.Ne geçmiÅŸ vardır,ne gelecek.Bunun mantıklı bir açıklkaması yok,ama herÅŸeyin bir mantığa dayanması gerekseydi,kim neyi neye dayayabilirdiki?AYRICAA;o adamın adı “aynÅŸtayn” deÄŸil adı “Einstein”.Ne biçim bi ansiklopedi bu böyle?Hepiniz(yorum kullananlar)ruhunuzu arıtme dersine mi gidiyorsunuz?herneyse,okulda çok zeki bi çocuk var.Ve yarına kadar bu bilgileri öğrenmem gerek.Åžans dileyin.Onu etkilemek istiyorum <33
Action requires knowledge, and now I can act!